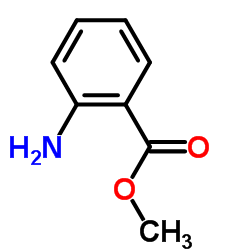
Methyl 2-Aminobenzoate
- CAS No.134-20-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Methyl 2-aminobenzoate (CAS 134-20-3), a key aromatic intermediate for fragrance and pharmaceutical synthesis.
Request Bulk PricingProduct Technical Details
Product Overview
Methyl 2-aminobenzoate, commonly known as Methyl anthranilate, is a versatile aromatic ester widely employed as a chemical intermediate in the synthesis of fine chemicals, fragrances, and active pharmaceutical ingredients. With a molecular formula of C8H9NO2 and a molecular weight of 151.16 g/mol, this compound exhibits distinctive blue fluorescence and is valued for its reactivity in electrophilic substitution and condensation reactions.
Specifications
| CAS Number | 134-20-3 |
|---|---|
| Molecular Formula | C8H9NO2 |
| Molecular Weight | 151.163 g/mol |
| Appearance | Colorless to light yellow liquid or crystalline solid with blue fluorescence |
| Assay (Purity) | ≥98% |
| Melting Point | 24 °C (lit.) |
| Boiling Point | 256.0 ± 0.0 °C at 760 mmHg |
| Density | 1.2 ± 0.1 g/cm³ |
| Flash Point | 104.4 ± 0.0 °C |
| LogP | 2.04 |
Industrial Applications
As a foundational building block in organic synthesis, Methyl 2-aminobenzoate serves critical roles across multiple industries:
- In the fragrance sector, it is used to formulate grape-like and fruity aroma compounds for perfumes, cosmetics, and flavoring agents.
- In pharmaceutical manufacturing, it acts as an intermediate in the production of heterocyclic compounds, including quinazolinones and benzodiazepine derivatives, supporting advanced organic synthesis pathways.
- It also finds application in agrochemical research and dye synthesis due to its electron-rich aromatic ring and bifunctional reactivity.
Our facility produces this intermediate under stringent quality control protocols, ensuring consistent batch-to-batch performance and compliance with international regulatory standards. Packaging is typically 220 kg per drum, with custom options available upon request. Store in a cool, dry, and well-ventilated area away from oxidizing agents.



