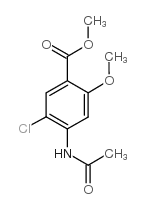
Methyl 4-acetamido-5-chloro-2-methoxybenzoate
- CAS No.4093-31-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used primarily in the synthesis of Metoclopramide hydrochloride. Available in bulk with comprehensive quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
Methyl 4-acetamido-5-chloro-2-methoxybenzoate is a specialized organic compound serving as a critical building block in the pharmaceutical industry. Identified by CAS number 4093-31-6, this substance functions primarily as a key intermediate in the manufacturing process of Metoclopramide hydrochloride, a widely recognized active pharmaceutical ingredient. Our facility ensures that every batch meets rigorous international standards for purity and consistency, supporting reliable downstream synthesis operations.
As a leading global manufacturer, we understand the importance of supply chain stability for pharmaceutical producers. This chemical is produced using advanced organic synthesis techniques that prioritize yield optimization and impurity control. The resulting product offers excellent solubility characteristics and reactivity profiles suitable for complex multi-step synthesis routes required in modern drug development.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 4093-31-6 |
| Molecular Formula | C11H12ClNO4 |
| Molecular Weight | 257.67 g/mol |
| Appearance | White powder |
| Purity (Assay) | ≥98.0% |
| Melting Point | 153-156 °C |
| Density | 1.312 g/cm3 |
| Boiling Point | 440.2 °C at 760 mmHg |
Quality Assurance and Control
Quality is the cornerstone of our manufacturing philosophy. Each production lot undergoes comprehensive analysis using high-performance liquid chromatography and other validated analytical methods. We provide a Certificate of Analysis (COA) with every shipment, detailing test results against our factory standards. Our internal quality control protocols ensure that parameters such as appearance, purity, and physical constants conform strictly to specified limits, minimizing risks for our partners during their formulation processes.
We maintain a robust quality management system that tracks materials from raw ingredient sourcing through final packaging. This traceability ensures that any potential issues can be identified and resolved swiftly. Our commitment to industrial purity means that customers receive a product ready for immediate use in sensitive pharmaceutical applications without requiring additional purification steps.
Applications and Usage
The primary application of this compound is in the synthesis of gastrointestinal therapeutic agents. Specifically, it serves as an essential precursor for Metoclopramide hydrochloride. Beyond this specific use, its chemical structure makes it a valuable candidate for research and development in related medicinal chemistry projects. Researchers utilizing this intermediate benefit from its consistent performance and reliable batch-to-batch reproducibility.
- Pharmaceutical API synthesis
- Medicinal chemistry research
- Custom organic synthesis projects
- Quality benchmark for intermediate standards
Packaging and Storage
To preserve product integrity during transit and storage, we utilize industry-standard packaging solutions. The standard packing configuration is 25 kg per drum, though we accommodate custom packaging requirements based on customer logistics needs. Proper storage involves keeping the material in a cool, ventilated environment away from direct sunlight and moisture. Adhering to these storage guidelines ensures the chemical stability and longevity of the product throughout its shelf life.
Our logistics team coordinates global shipping to ensure timely delivery while maintaining compliance with international chemical transport regulations. We support bulk orders and provide competitive pricing structures for long-term partnerships. For detailed technical data or procurement inquiries, our sales team is available to provide comprehensive support and documentation.



