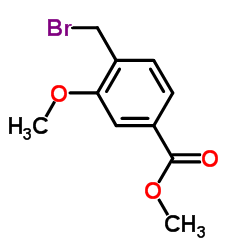
Methyl 4-(bromomethyl)-3-methoxybenzoate
- CAS No.70264-94-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity pharmaceutical intermediate used primarily in the synthesis of Zafirlukast. Available in bulk quantities with comprehensive quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
Methyl 4-(bromomethyl)-3-methoxybenzoate is a specialized organic compound widely recognized for its critical role in pharmaceutical manufacturing processes. This chemical building block serves as a key precursor in the synthesis of advanced therapeutic agents, specifically functioning as an essential intermediate in the production of Zafirlukast. Our facility produces this compound under strict quality control standards to ensure consistency and reliability for downstream pharmaceutical applications.
The molecular structure features a benzoate ester backbone with specific methoxy and bromomethyl substitutions, providing unique reactivity profiles suitable for complex organic synthesis. This compound is designed for industrial-scale usage where high purity and precise physicochemical properties are paramount for successful reaction outcomes.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 70264-94-7 |
| Molecular Formula | C10H11BrO3 |
| Molecular Weight | 259.096 g/mol |
| Appearance | White to off-white crystalline powder |
| Assay | ≥98.0% |
| Melting Point | 92-94 °C |
| Boiling Point | 324.2 °C at 760 mmHg |
| Density | 1.4 ± 0.1 g/cm3 |
| Flash Point | 149.9 °C |
| Refractive Index | 1.547 |
Industrial Applications
The primary application of this intermediate lies within the pharmaceutical sector, specifically supporting the manufacturing pipeline for leukotriene receptor antagonists. Its high reactivity allows for efficient coupling and substitution reactions required in multi-step synthesis routes. Beyond its specific use in Zafirlukast production, this chemical serves as a versatile building block for various organic synthesis projects requiring functionalized aromatic esters.
- Essential precursor for asthma medication synthesis
- Compatible with standard organic reaction conditions
- Supports scalable manufacturing processes
- High stability under recommended storage conditions
Quality Assurance and Packaging
We maintain rigorous quality assurance protocols to guarantee that every batch meets or exceeds the specified assay requirements. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) detailing physical and chemical test results. Our production facility adheres to international safety and environmental standards, ensuring responsible manufacturing practices.
Standard packaging consists of 25 kg drums designed to protect the product from moisture and light exposure during transit. Custom packaging solutions are available upon request to accommodate specific logistical requirements. We recommend storing this material in a cool, ventilated area away from incompatible substances to maintain optimal stability and shelf life. Our global supply chain ensures timely delivery to support continuous manufacturing operations worldwide.



