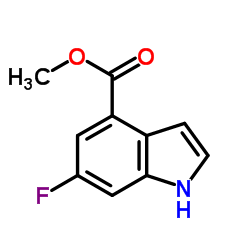
Methyl 6-fluoro-1H-indole-4-carboxylate
- CAS No.1082040-43-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Methyl 6-fluoro-1H-indole-4-carboxylate (CAS 1082040-43-4) designed for pharmaceutical synthesis. Ideal intermediate for oncology research and API manufacturing with verified quality standards.
Request Bulk PricingProduct Technical Details
Product Overview
Methyl 6-fluoro-1H-indole-4-carboxylate is a specialized pharmaceutical intermediate characterized by its fluorinated indole structure. This compound serves as a critical building block in the synthesis of advanced therapeutic agents, particularly within the oncology sector. Our manufacturing process ensures high chemical integrity, making it suitable for complex organic synthesis routes required in modern drug development.
As a key precursor, this fluorinated ester facilitates the construction of poly-ADP-ribose polymerase (PARP) inhibitor pathways. The presence of the fluoro substituent enhances metabolic stability and binding affinity in downstream applications. We maintain strict quality control protocols to guarantee consistency across batches, supporting both research laboratories and large-scale production facilities.
Technical Specifications
| Parameter | Value |
|---|---|
| Chemical Name | Methyl 6-fluoro-1H-indole-4-carboxylate |
| CAS Number | 1082040-43-4 |
| Molecular Formula | C10H8FNO2 |
| Molecular Weight | 193.17 g/mol |
| Purity (HPLC) | ≥98.0% |
| Appearance | White to yellow powder |
Industrial Applications
This intermediate is primarily utilized in the manufacturing process of targeted cancer therapies. It acts as a foundational element for constructing complex heterocyclic systems found in novel anticancer drugs. Researchers and process chemists value this compound for its reactivity profile and compatibility with various coupling reactions.
- Essential building block for PARP inhibitor synthesis
- Used in medicinal chemistry for structure-activity relationship studies
- Supports scale-up processes for clinical trial material production
- Compatible with standard organic synthesis methodologies
Quality Assurance and Packaging
We adhere to rigorous quality management systems to ensure product reliability. Each batch is accompanied by a Certificate of Analysis (COA) detailing purity levels and impurity profiles. Our facility employs advanced analytical techniques, including HPLC and NMR, to verify chemical identity and potency.
Standard packaging consists of 25 kg drums, designed to maintain stability during transit. Custom packaging solutions are available upon request to meet specific logistical requirements. We recommend storing the product in a cool, ventilated area away from direct sunlight and moisture to preserve shelf life. Our global supply chain ensures timely delivery to qualified pharmaceutical manufacturers and research institutions worldwide.



