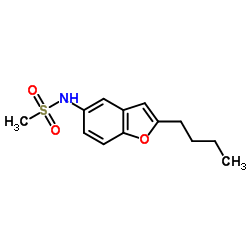
N-(2-Butylbenzofuran-5-yl)methanesulfonamide
- CAS No.437652-07-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity N-(2-Butylbenzofuran-5-yl)methanesulfonamide for Dronedarone synthesis. Reliable bulk supply with full COA and competitive pricing for pharmaceutical manufacturers.
Request Bulk PricingProduct Technical Details
Product Overview
N-(2-Butylbenzofuran-5-yl)methanesulfonamide is a specialized pharmaceutical intermediate characterized by its high chemical stability and precise molecular structure. This compound serves as a critical building block in the synthesis of advanced cardiovascular medications, specifically acting as a key precursor in the production of Dronedarone hydrochloride. Our manufacturing facility employs state-of-the-art organic synthesis techniques to ensure consistent quality and purity levels that meet stringent international pharmaceutical standards.
The chemical identity of this substance is defined by its specific benzofuran sulfonamide derivative structure, which facilitates efficient downstream reactions. We understand the critical nature of supply chain reliability for active pharmaceutical ingredient (API) synthesis. Therefore, our production lines are optimized for scalability, allowing us to meet both pilot-scale requirements and large-scale commercial manufacturing demands without compromising on quality parameters.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 437652-07-8 |
| Molecular Formula | C13H17NO3S |
| Molecular Weight | 267.34 g/mol |
| Appearance | Off-white powder |
| Purity | ≥98.0% |
| Moisture Content | ≤0.1% |
| Total Impurities | ≤0.5% |
| Melting Point | 104-106°C |
| Boiling Point | 399.86°C at 760 mmHg |
Industrial Applications
The primary application of N-(2-Butylbenzofuran-5-yl)methanesulfonamide lies within the pharmaceutical sector, specifically targeting the cardiovascular therapeutic market. It functions as an essential intermediate in the multi-step synthesis route of Dronedarone, an antiarrhythmic medication used to maintain normal heart rhythm. The high purity grade we supply ensures minimal byproduct formation during subsequent coupling reactions, thereby improving overall yield and reducing purification costs for our clients.
Beyond its direct use in Dronedarone production, this benzofuran derivative demonstrates versatility in medicinal chemistry research. It serves as a valuable reference standard for analytical method development and quality control testing. Our technical team supports customers with detailed synthesis route documentation and manufacturing process insights to facilitate seamless integration into your existing production workflows.
Quality Assurance and Storage
- Strict quality control protocols implemented at every stage of production.
- Comprehensive Certificate of Analysis (COA) provided with every batch.
- Manufactured under GMP standards to ensure regulatory compliance.
- Available for custom synthesis requests based on specific purity requirements.
- Global shipping capabilities with secure packaging to prevent degradation.
Proper storage is essential to maintain the integrity of this chemical intermediate. We recommend storing the material in a cool, dry, and well-closed container, protected from moisture and strong light or heat sources. Our standard packaging includes 25kg cardboard drums, though we accommodate custom packaging solutions based on client specifications. By choosing our facility as your global manufacturer, you gain access to competitive bulk pricing, reliable lead times, and a partnership focused on long-term supply security for your pharmaceutical synthesis needs.



