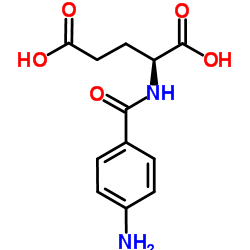
N-(4-Aminobenzoyl)-L-glutamic acid
- CAS No.4271-30-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate essential for Folic Acid and Methotrexate synthesis, available in bulk quantities with comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
N-(4-Aminobenzoyl)-L-glutamic acid, frequently recognized within the pharmaceutical industry as Folic Acid Impurity A or 4-Aminobenzoylglutamic acid, stands as a vital building block in the synthesis of complex therapeutic agents. This specialized organic compound plays a pivotal role in the manufacturing pathways of antifolate medications and vitamin supplements. Our facility produces this intermediate under strict quality control protocols to ensure consistency and reliability for downstream pharmaceutical applications.
As a key precursor, this compound is integral to the production of essential medicines used in oncology and nutritional health. The molecular structure, characterized by the conjugation of 4-aminobenzoic acid with L-glutamic acid, requires precise stereochemical control during synthesis. We leverage advanced organic synthesis techniques to maintain high optical purity and chemical stability, ensuring that every batch meets the rigorous demands of modern drug manufacturing.
Technical Specifications
Our production process is designed to deliver superior purity levels suitable for sensitive pharmaceutical formulations. The following table outlines the critical quality attributes verified through our analytical laboratories:
| Parameter | Specification | Typical Result |
|---|---|---|
| Appearance | White crystalline powder | Conform |
| CAS Number | 4271-30-1 | 4271-30-1 |
| Molecular Formula | C12H14N2O5 | C12H14N2O5 |
| Molecular Weight | 266.25 g/mol | 266.25 g/mol |
| Melting Point | 168-174℃ | 172.8-173.4℃ |
| Assay (HPLC) | ≥98.0% | 99.89% |
| Loss on Drying | ≤2.0% | 0.34% |
| Residue on Ignition | ≤3.0% | 1.10% |
Industrial Applications
The primary application of N-(4-Aminobenzoyl)-L-glutamic acid lies in the pharmaceutical sector, specifically serving as a crucial intermediate for the synthesis of Methotrexate and Folic Acid. These end products are fundamental in treating various medical conditions, ranging from vitamin deficiencies to autoimmune disorders and certain types of cancer. By providing a high-quality starting material, we support the global supply chain for these essential medicines.
Beyond direct pharmaceutical synthesis, this compound is utilized in research and development settings for studying folate metabolism pathways. Its high purity ensures accurate results in biochemical assays and clinical trial material production. Manufacturers rely on our consistent supply to maintain their production schedules and meet regulatory compliance standards without interruption.
Quality Assurance and Storage
We understand that the integrity of pharmaceutical intermediates is non-negotiable. Every batch of N-(4-Aminobenzoyl)-L-glutamic acid is accompanied by a comprehensive Certificate of Analysis (COA), detailing test results against factory standards. Our quality management system ensures traceability from raw material sourcing to final packaging.
To maintain optimal stability, the product should be stored in a cool, dry environment. The recommended storage condition is at -20℃ to prevent degradation over extended periods. Proper handling procedures should be followed to ensure safety and maintain the chemical properties of the crystalline powder. We offer flexible packaging options to suit both laboratory-scale research and large-scale industrial manufacturing needs.



