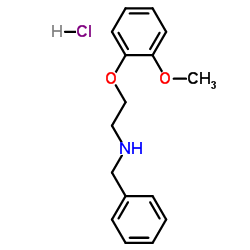
N-Benzyl-2-(2-methoxyphenoxy)ethanamine
- CAS No.3246-03-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity N-Benzyl-2-(2-methoxyphenoxy)ethanamine (CAS 3246-03-5) designed for pharmaceutical synthesis. Ideal intermediate for Carvedilol production with ≥98% purity assurance.
Request Bulk PricingProduct Technical Details
Product Overview
Our company specializes in the production and supply of high-quality pharmaceutical intermediates, including N-Benzyl-2-(2-methoxyphenoxy)ethanamine. This chemical compound serves as a critical building block in the synthesis of advanced cardiovascular medications. Manufactured under strict quality control protocols, this intermediate ensures consistent performance in downstream pharmaceutical applications. We understand the rigorous demands of the global pharmaceutical supply chain and deliver products that meet international standards for purity and safety.
As a leading manufacturer, we prioritize the structural integrity and chemical stability of our intermediates. N-Benzyl-2-(2-methoxyphenoxy)ethanamine is synthesized using optimized routes to minimize impurities and maximize yield. This commitment to excellence makes our product a preferred choice for pharmaceutical companies seeking reliable raw materials for large-scale production. Our facility is equipped with state-of-the-art analytical instruments to verify every batch before shipment.
Key Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 3246-03-5 |
| Molecular Formula | C16H20ClNO2 |
| Molecular Weight | 293.79 g/mol |
| Appearance | White crystalline powder |
| Purity | ≥98% |
| Single Impurity | ≤1.0% |
| Total Impurities | ≤2.0% |
| Density | 1.072±0.06 g/cm3 |
| Boiling Point | 160-161 °C |
Industrial Applications
The primary application of N-Benzyl-2-(2-methoxyphenoxy)ethanamine is in the pharmaceutical industry, specifically as a key intermediate in the synthesis of Carvedilol and Carvedilol phosphate. Carvedilol is a widely prescribed non-selective beta-blocker used to treat high blood pressure and heart failure. The quality of the intermediate directly impacts the efficacy and safety of the final drug product. Therefore, maintaining high purity levels and consistent physicochemical properties is essential for regulatory compliance and patient safety.
Our intermediate supports efficient synthesis routes, reducing reaction times and improving overall yield for our clients. By providing a stable and high-purity starting material, we help pharmaceutical manufacturers streamline their production processes. This compound is also utilized in research and development settings for creating novel analogues and derivatives within the cardiovascular therapeutic class. Our technical team is available to provide support on integration into various synthetic pathways.
Quality Assurance and Packaging
- Every batch undergoes rigorous testing using HPLC and GC methods to confirm purity levels.
- Certificate of Analysis (COA) is provided with each shipment for full traceability.
- Packaged in 25kg cardboard drums to ensure protection during transit.
- Custom packaging solutions are available based on specific customer requirements.
- Stored in cool, dry, well-closed containers away from moisture and strong light.
We maintain a robust supply chain to ensure timely delivery to global destinations. Our storage facilities adhere to strict environmental controls to prevent degradation of the chemical structure. Safety data sheets (SDS) are available upon request to ensure safe handling and compliance with local regulations. Partner with us for a reliable source of pharmaceutical intermediates that meet your exacting standards for quality and performance.



