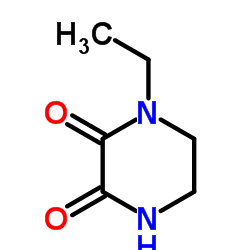
N-Ethyl-2,3-dioxopiperazine
- CAS No.59702-31-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used primarily in the synthesis of Cefoperazone. Available in bulk with strict quality control and comprehensive documentation.
Request Bulk PricingProduct Technical Details
Product Overview
N-Ethyl-2,3-dioxopiperazine, identified by CAS number 59702-31-7, is a specialized organic compound serving as a critical building block in the pharmaceutical industry. This piperazine derivative is meticulously synthesized to meet stringent quality standards required for active pharmaceutical ingredient (API) production. Our facility ensures that every batch undergoes rigorous analytical testing to guarantee consistency and reliability for downstream synthesis processes.
As a key precursor, this compound plays an indispensable role in the manufacturing of cephalosporin antibiotics. The chemical structure features a stable dioxopiperazine ring with an ethyl substitution, providing the necessary reactivity and stability for complex organic transformations. Our commitment to excellence ensures that clients receive material that facilitates efficient reaction yields and minimizes purification challenges.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | N-Ethyl-2,3-dioxopiperazine |
| CAS Number | 59702-31-7 |
| Molecular Formula | C6H10N2O2 |
| Molecular Weight | 142.16 g/mol |
| Appearance | White or off-white powder |
| Purity | ≥98.0% |
| Melting Point | 106-110 °C |
| Moisture Content | ≤0.5% |
| Total Impurities | ≤0.1% |
Industrial Applications
The primary application of N-Ethyl-2,3-dioxopiperazine is as a vital intermediate in the synthesis of Cefoperazone, a third-generation cephalosporin antibiotic. In this context, the purity of the intermediate directly influences the quality and safety profile of the final medicinal product. Our manufacturing process is optimized to reduce specific impurities that could complicate the final drug substance purification.
Beyond antibiotic synthesis, this chemical building block is utilized in various research and development projects involving heterocyclic chemistry. Laboratories engaged in the development of new neurological therapeutics or advanced organic synthesis protocols may require this compound for structure-activity relationship studies. The consistent physical properties, including density and refractive index, make it a reliable standard for comparative analysis.
Quality Assurance and Packaging
We adhere to strict quality management systems to ensure product integrity from synthesis to shipment. Each production lot is accompanied by a Certificate of Analysis (COA) detailing all critical quality attributes. Our quality control team employs high-performance liquid chromatography (HPLC) and nuclear magnetic resonance (NMR) spectroscopy to verify chemical identity and purity levels.
Standard packaging consists of 25kg cardboard drums designed to protect the material from environmental factors. Custom packaging solutions are available upon request to accommodate specific logistical requirements. We prioritize safety and compliance, ensuring that all labeling meets international hazardous materials transportation regulations where applicable.
Storage and Handling
To maintain optimal stability, N-Ethyl-2,3-dioxopiperazine should be stored in a cool, dry, and well-closed container. It is essential to keep the material away from moisture, strong light, and excessive heat, as these factors can degrade chemical quality over time. Proper handling procedures include using appropriate personal protective equipment (PPE) to prevent inhalation or skin contact during transfer operations.
Our global supply chain capabilities ensure timely delivery to manufacturers worldwide. Whether you require small-scale quantities for research or bulk volumes for commercial production, we are equipped to meet your demand with competitive pricing and reliable service. Contact our technical sales team for detailed information regarding synthesis routes, safety data sheets, and current availability.



