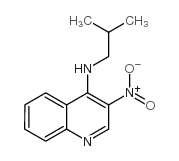
N-Isobutyl-3-nitroquinolin-4-amine
- CAS No.99009-85-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used in the synthesis of Imiquimod, available in bulk quantities with full quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
N-Isobutyl-3-nitroquinolin-4-amine is a specialized organic compound primarily utilized within the pharmaceutical industry as a critical building block. This quinoline derivative serves as a key precursor in the multi-step synthesis of active pharmaceutical ingredients, specifically functioning as an intermediate for Imiquimod production. Our manufacturing facility ensures that every batch meets rigorous international standards for chemical purity and consistency, supporting reliable downstream processing for our global partners.
As a seasoned manufacturer with decades of experience in fine chemical synthesis, we understand the importance of molecular integrity in drug development. This compound features a distinct nitroquinoline structure coupled with an isobutyl amine group, requiring precise control during production to maintain stability and reactivity. We employ advanced chromatographic purification techniques to eliminate trace impurities, ensuring the material is suitable for sensitive pharmaceutical applications.
Physical And Chemical Properties
The structural characteristics of this amine derivative contribute to its specific reactivity profile. Understanding these properties is essential for process chemists when designing synthesis routes or scaling up production. The compound exhibits stable thermal properties within standard storage conditions, though care must be taken to avoid exposure to strong oxidizing agents. The molecular weight and density values provided are based on standard theoretical calculations and verified through empirical measurement in our quality control laboratory.
| Property | Value |
|---|---|
| CAS Number | 99009-85-5 |
| Molecular Formula | C13H15N3O2 |
| Molecular Weight | 245.28 g/mol |
| Density | 1.239 g/cm3 |
| Boiling Point | 398.022 °C at 760 mmHg |
| Flash Point | 194.517 °C |
| Index Of Refraction | 1.65 |
Quality Specifications
Quality assurance is the cornerstone of our manufacturing process. Each production lot undergoes comprehensive testing using High-Performance Liquid Chromatography (HPLC) and Nuclear Magnetic Resonance (NMR) spectroscopy. We guarantee a minimum assay purity that exceeds industry norms, minimizing the risk of side reactions in subsequent synthesis steps. Our commitment to transparency means that a Certificate of Analysis (COA) is provided with every shipment, detailing batch-specific data.
- Appearance: Yellow crystalline powder
- Assay: Greater than or equal to 99.0 percent
- Single Impurity: Less than or equal to 0.2 percent
- Total Impurities: Less than or equal to 1.0 percent
Industrial Applications
The primary application of N-Isobutyl-3-nitroquinolin-4-amine lies in the pharmaceutical sector. It is predominantly used as an intermediate in the manufacture of Imiquimod, an immune response modifier used in dermatological treatments. Beyond this specific pathway, the quinoline scaffold offers versatility for medicinal chemistry research, potentially serving as a template for developing novel heterocyclic compounds. Our R&D team supports clients with technical data regarding solubility and reaction conditions to facilitate efficient process integration.
Packaging And Storage
To preserve chemical integrity during transit, we utilize industry-standard packaging solutions. The default configuration is 25 kg drums lined with protective polyethylene bags to prevent moisture absorption and contamination. However, we offer flexible packaging options tailored to customer requirements, including smaller quantities for laboratory scale-up or larger bulk containers for industrial production. Storage should be maintained in a cool, ventilated area away from direct sunlight and incompatible materials. Proper handling procedures, including the use of personal protective equipment, are recommended during transfer operations.



