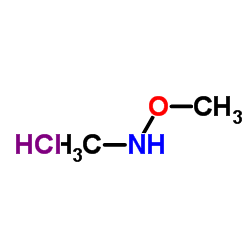
N,O-Dimethylhydroxylamine Hydrochloride
- CAS No.6638-79-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity N,O-Dimethylhydroxylamine hydrochloride for efficient Weinreb amide synthesis. Ideal for pharmaceutical and pesticide intermediate manufacturing.
Request Bulk PricingProduct Technical Details
Product Overview
N,O-Dimethylhydroxylamine hydrochloride stands as a fundamental reagent in the realm of advanced organic synthesis, particularly valued for its indispensable role in the construction of Weinreb amides. This specialized chemical building block enables chemists to achieve precise control over carbonyl formation, facilitating the conversion of acid chlorides into ketones without the risk of over-addition that typically occurs with standard organometallic reagents. Our production facility is dedicated to supplying this critical intermediate with the highest levels of consistency and purity, supporting both research laboratories and large-scale industrial manufacturing operations.
The hydrochloride salt form provides significant advantages in terms of stability and handling safety compared to the free base variant. This makes it an ideal choice for processes requiring robust storage conditions and reliable performance across extended periods. We understand that the quality of starting materials directly impacts the yield and purity of final pharmaceutical or agrochemical products. Therefore, our manufacturing protocols are designed to minimize impurities and ensure batch-to-batch reproducibility, adhering to international quality standards.
Technical Specifications
| Chemical Name | N,O-Dimethylhydroxylamine Hydrochloride |
|---|---|
| CAS Registry Number | 6638-79-5 |
| Molecular Formula | C2H8ClNO |
| Molecular Weight | 97.54 g/mol |
| Physical Appearance | White to Light Yellow Crystalline Powder |
| Assay Purity | ≥98.0% |
| Moisture Content | ≤0.5% |
| Melting Point Range | 112-115°C |
Industrial Applications
The primary utility of N,O-Dimethylhydroxylamine hydrochloride is found in the preparation of N-methoxy-N-methylamides, commonly referred to as Weinreb amides. These intermediates are crucial in modern medicinal chemistry for the synthesis of complex ketone structures. The unique chelating ability of the methoxy methyl amide moiety allows for the stable coordination of organolithium or Grignard reagents, effectively halting the reaction at the ketone stage. This specificity is vital for the development of active pharmaceutical ingredients where structural precision is paramount.
- Pharmaceutical Intermediate Synthesis: Extensively utilized in the multi-step synthesis of APIs requiring specific carbonyl functionalization.
- Agrochemical Development: Serves as a key precursor in the creation of novel pesticide molecules with enhanced biological activity.
- Fine Chemical Manufacturing: Employed in the production of specialized organic compounds for material science and research applications.
Quality Assurance and Storage
Quality control is the cornerstone of our supply chain. Every batch of N,O-Dimethylhydroxylamine hydrochloride undergoes rigorous analytical testing using high-performance liquid chromatography (HPLC) and nuclear magnetic resonance (NMR) spectroscopy. These methods confirm chemical identity and quantify purity levels to ensure compliance with specified parameters. We provide comprehensive documentation, including Certificates of Analysis (COA), to support your regulatory filings and quality assurance audits.
For optimal preservation of product integrity, storage in a cool, dry, and well-ventilated environment is recommended. The material should be kept away from direct sunlight and sources of heat to prevent degradation. Containers must be tightly sealed to avoid moisture absorption, as the compound is hygroscopic. We offer flexible packaging options, typically supplying in 25 kg drums, but can accommodate custom packaging requests to suit specific logistical needs. Our commitment to safety and quality ensures that you receive a reliable product ready for immediate integration into your synthesis workflows.



