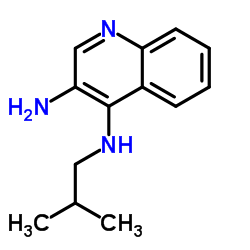
N4-Isobutylquinoline-3,4-diamine
- CAS No.99010-09-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used in the synthesis of immune response modifiers. Available in bulk quantities with full documentation.
Request Bulk PricingProduct Technical Details
Product Overview
N4-Isobutylquinoline-3,4-diamine is a specialized organic compound primarily utilized within the pharmaceutical industry as a critical building block. This quinoline derivative serves as a key intermediate in the multi-step synthesis of advanced immunomodulatory agents. Our facility produces this chemical under strict quality control protocols to ensure consistency and reliability for downstream pharmaceutical manufacturing processes.
As a regulated chemical entity, this product is designed for professional laboratory and industrial use only. It represents a vital component in the supply chain for developing therapeutic compounds that target immune system modulation. We prioritize safety and compliance in every batch produced.
Chemical Properties and Structure
The molecular structure of this diamine features a quinoline core substituted with an isobutyl amino group. This specific configuration is essential for its reactivity in subsequent coupling reactions. The compound exhibits stable physical characteristics suitable for long-term storage when handled according to recommended guidelines. Its chemical stability makes it an ideal candidate for complex organic synthesis routes requiring precise stoichiometry.
Understanding the physical constants is crucial for process engineers. The density and boiling point data provided allow for accurate calculation of reaction parameters. Our technical team ensures that every shipment matches the specified physical profile, minimizing variability in your production line.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 99010-09-0 |
| Molecular Formula | C13H17N3 |
| Molecular Weight | 215.29 g/mol |
| Appearance | Light yellow crystalline powder |
| Purity | ≥98.0% |
| Water Content | ≤0.5% |
| Boiling Point | 379.6°C at 760 mmHg |
| Flash Point | 183.4°C |
Industrial Applications
This intermediate is predominantly employed in the synthesis of pharmacologically active compounds related to immune response modifiers. It acts as a precursor in the construction of molecules that stimulate interferon production. Research and development teams utilize this material to explore new therapeutic avenues in dermatology and oncology support care.
Manufacturers rely on consistent quality to maintain regulatory compliance for their final drug products. Our supply chain is optimized to deliver this intermediate with the necessary documentation to support your regulatory filings. We understand the critical nature of raw material consistency in pharmaceutical manufacturing.
Quality Assurance and Safety
Every batch undergoes rigorous analytical testing using high-performance liquid chromatography and mass spectrometry. We provide a comprehensive Certificate of Analysis with each order to verify purity and identity. Our quality management system adheres to international standards to ensure product safety and efficacy.
Proper handling is essential for maintaining product integrity. Users should employ standard laboratory safety practices including the use of personal protective equipment. Storage conditions must be maintained to prevent degradation over time. We recommend consulting the safety data sheet for detailed handling instructions before use.
Packaging and Logistics
Standard packaging consists of secure cardboard drums containing 25 kilograms of material. Custom packaging solutions are available upon request to meet specific logistical requirements. We offer global shipping services with careful attention to regulatory compliance for chemical transport. Our logistics partners ensure timely delivery while maintaining product stability during transit.



