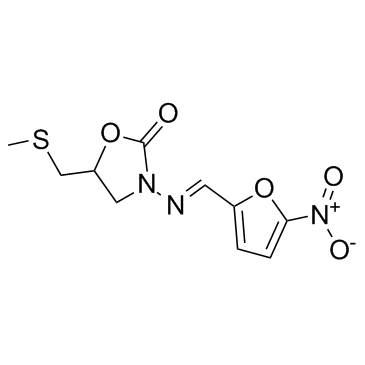
Nifuratel
- CAS No.4936-47-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Nifuratel API (CAS 4936-47-4) supplied for pharmaceutical formulation. Meets strict pharmacopoeia standards for antimicrobial applications.
Request Bulk PricingProduct Technical Details
Product Overview
Nifuratel is a specialized pharmaceutical intermediate and active pharmaceutical ingredient (API) widely recognized for its broad-spectrum antimicrobial properties. Manufactured under strict quality control protocols, this compound serves as a critical component for licensed pharmaceutical companies developing therapeutic formulations. Our facility ensures that every batch meets rigorous international standards for purity, safety, and consistency, making it an ideal choice for large-scale drug production.
Chemically known as 5-(methylthiomethyl)-3-((5-nitrofuran-2-yl)methyleneamino)oxazolidin-2-one, this substance exhibits stable physicochemical characteristics suitable for various dosage forms. The molecular structure provides robust activity profiles required for specific medical applications, while our advanced synthesis routes guarantee minimal impurity levels. We prioritize supply chain reliability to support continuous manufacturing operations for our global partners.
Technical Specifications
Quality assurance is paramount in pharmaceutical manufacturing. Our Nifuratel undergoes comprehensive analytical testing to verify compliance with factory and pharmacopoeia standards. Key parameters such as assay purity, residual solvents, and related substances are meticulously monitored. The following table outlines the typical specification profile for our standard production batches:
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | Yellow crystalline powder | Yellow crystalline powder |
| pH | 5.5-7.0 | 6.23 |
| Colour of solution | No darker than No.5 standard solution | Conform |
| Melting point | 186-189℃ | 187.5-188.5℃ |
| Residual Solvents (DMF) | ≤880ppm | ND |
| Residual Solvents (Methanol) | ≤3000ppm | ND |
| Related substances (Total) | ≤0.5% | 0.16% |
| Loss on drying | ≤0.5% | 0.08% |
| Assay | 98.5-101.5% | 100.7% |
Industrial Applications
This chemical is primarily utilized in the formulation of antimicrobial medications intended for professional healthcare use. Due to its efficacy against a range of pathogens, it is a valuable asset for companies producing treatments targeting specific bacterial and protozoal infections. Our product is designed as a drop-in replacement for existing supply chains, offering consistent performance benchmarks for formulation scientists. We support clients in optimizing their manufacturing processes with high-quality raw materials that reduce downstream processing risks.
Quality Assurance and Storage
To maintain integrity, proper storage conditions are essential. The material should be kept in a cool, dry environment, ideally at freezer temperatures around -20°C, to prevent degradation over time. Each shipment is accompanied by a Certificate of Analysis (COA) detailing batch-specific test results. Our commitment to quality extends beyond production, encompassing secure packaging and logistics solutions that ensure the product arrives in optimal condition. Partner with us for reliable bulk pricing and a global manufacturing network dedicated to pharmaceutical excellence.



