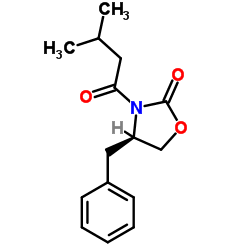
(R)-3-(3-Methylbutanoyl)-4-benzyloxazolidin-2-one
- CAS No.145589-03-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity chiral intermediate for Aliskiren synthesis. Manufactured under strict GMP standards with ≥98% purity assurance.
Request Bulk PricingProduct Technical Details
Product Overview
(R)-3-(3-Methylbutanoyl)-4-benzyloxazolidin-2-one is a specialized chiral building block essential for the pharmaceutical industry. This oxazolidinone derivative serves as a critical precursor in the stereoselective synthesis of complex therapeutic agents. Our facility produces this compound with rigorous attention to stereochemical integrity, ensuring consistent performance in downstream API manufacturing processes.
As a key intermediate, this compound facilitates the construction of specific chiral centers required for biological activity in final drug products. The robust chemical structure provides stability during various synthetic transformations, making it a reliable choice for process chemists and procurement managers alike.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 145589-03-3 |
| Molecular Formula | C15H19NO3 |
| Molecular Weight | 261.32 g/mol |
| Purity | ≥98% |
| Appearance | White powder |
| Density | 1.152 g/cm3 |
| Boiling Point | 412.9ºC at 760mmHg |
| Flash Point | 203.5ºC |
Industrial Applications
This chemical is primarily utilized as a vital intermediate in the synthesis of Aliskiren and its hemifumarate salt. Aliskiren is a well-known direct renin inhibitor used in the management of hypertension. The high optical purity of our (R)-3-(3-Methylbutanoyl)-4-benzyloxazolidin-2-one ensures that the final API meets stringent regulatory requirements for enantiomeric excess.
Beyond cardiovascular applications, oxazolidinone scaffolds are increasingly explored in other therapeutic areas. Our manufacturing capabilities support custom synthesis routes tailored to specific project needs, allowing for scalability from laboratory research to commercial production volumes.
Quality Assurance and Safety
We adhere to comprehensive quality management systems to guarantee product consistency. Every batch is accompanied by a Certificate of Analysis (COA) detailing critical quality attributes. Our production environment maintains strict control over moisture and temperature to preserve chemical stability.
- Strict GMP-compliant manufacturing processes
- Advanced analytical testing for purity verification
- Secure packaging to prevent degradation during transit
- Global logistics support for timely delivery
Storage and Handling
To maintain optimal quality, store this material in a cool, dry, and well-closed container. Protect from moisture and strong light or heat sources. Proper handling procedures should be followed according to standard laboratory safety protocols. We recommend consulting the Safety Data Sheet (SDS) for detailed handling instructions before use.



