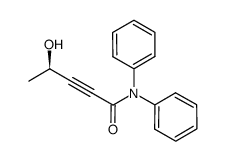
(R)-4-Hydroxy-N,N-diphenylpent-2-ynamide
- CAS No.899809-61-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity chiral intermediate essential for the synthesis of advanced pharmaceutical compounds. Manufactured under strict quality control standards for global distribution.
Request Bulk PricingProduct Technical Details
Product Overview
(R)-4-Hydroxy-N,N-diphenylpent-2-ynamide is a specialized chiral pharmaceutical intermediate designed for high-level organic synthesis applications. This compound serves as a critical building block in the manufacturing process of complex therapeutic agents, specifically acting as a key precursor in the production of Vorapaxar Sulfate. Our facility ensures that every batch meets rigorous international standards for purity and stereochemical integrity, making it an ideal choice for research and development teams as well as large-scale production units.
The molecular structure features a distinct hydroxy group and diphenylamide functionality, which are crucial for the subsequent chemical transformations required in downstream synthesis. Maintaining the correct (R)-configuration is vital for the biological activity of the final pharmaceutical product. We employ advanced chromatographic techniques and spectroscopic analysis to verify the enantiomeric excess and chemical purity of each lot before release.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 899809-61-1 |
| Molecular Formula | C17H15NO2 |
| Molecular Weight | 265.31 g/mol |
| Appearance | White powder |
| Assay | ≥98.0% |
| Density | 1.215±0.06 g/cm3 |
| Residue on Ignition | ≤0.5% |
Quality Assurance and Control
Quality is the cornerstone of our manufacturing philosophy. Each production run of (R)-4-Hydroxy-N,N-diphenylpent-2-ynamide undergoes a multi-stage verification process. This includes raw material screening, in-process control checks, and final product validation using High-Performance Liquid Chromatography (HPLC) and Nuclear Magnetic Resonance (NMR) spectroscopy. We provide a comprehensive Certificate of Analysis (COA) with every shipment, detailing all critical quality attributes and test results.
Our production facilities adhere to strict safety and environmental guidelines. We utilize state-of-the-art equipment to minimize contamination risks and ensure consistent batch-to-batch reproducibility. This commitment to quality allows our partners to streamline their own regulatory filings and production schedules with confidence in the raw materials supplied.
Industrial Applications
Primarily utilized as an intermediate in the pharmaceutical industry, this compound is essential for the synthesis of platelet aggregation inhibitors. Its high purity profile ensures efficient reaction yields in subsequent steps, reducing waste and optimizing overall process economics. Beyond its primary use, it may also serve as a reference standard or a building block in the development of novel organic compounds requiring similar structural motifs.
Packaging and Storage
To maintain stability and integrity during transit, the product is packaged in secure 25 kg drums. Custom packaging solutions are available upon request to meet specific logistical requirements. Storage recommendations include keeping the material in a cool, ventilated area away from direct sunlight and moisture. Proper handling procedures should be followed to ensure safety and preserve the chemical properties of the substance throughout its shelf life.
Global Supply Chain
We understand the importance of timely delivery in the pharmaceutical supply chain. Our logistics network supports global shipping, ensuring that orders are processed efficiently and delivered safely to destinations worldwide. Whether you require sample quantities for evaluation or bulk volumes for commercial manufacturing, our team is equipped to handle your requirements with professionalism and speed.



