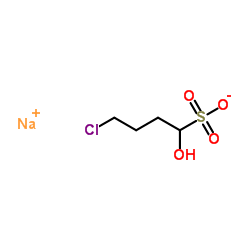
Sodium 4-Chloro-1-Hydroxybutane-1-Sulfonate
- CAS No.54322-20-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Sodium 4-Chloro-1-Hydroxybutane-1-Sulfonate (CAS 54322-20-2) for pharmaceutical synthesis. Reliable supply for global API manufacturing.
Request Bulk PricingProduct Technical Details
Product Overview
Sodium 4-Chloro-1-Hydroxybutane-1-Sulfonate is a critical organic intermediate utilized extensively in the pharmaceutical industry for the synthesis of complex therapeutic agents. This specialized chemical compound serves as a foundational building block in the production of various migraine treatment medications, ensuring high efficiency and reliability in downstream manufacturing processes. Our facility adheres to strict quality control protocols to deliver material that meets rigorous international standards for pharmaceutical-grade intermediates.
As a leading global manufacturer, we understand the importance of consistency and purity in chemical supply chains. This product is engineered to facilitate smooth reaction pathways, minimizing impurities that could affect the final active pharmaceutical ingredient (API) quality. The compound is characterized by its stability and reactivity, making it an ideal choice for large-scale industrial synthesis operations.
Technical Specifications
Our production process ensures that every batch meets or exceeds the specified purity requirements. We employ advanced analytical techniques to verify composition and physical properties, providing customers with comprehensive documentation including Certificates of Analysis (COA) for every shipment. The following table outlines the key technical parameters for this intermediate:
| Parameter | Specification |
|---|---|
| Chemical Name | Sodium 4-Chloro-1-Hydroxybutane-1-Sulfonate |
| CAS Number | 54322-20-2 |
| Molecular Formula | C4H8ClNaO4S |
| Molecular Weight | 210.61 g/mol |
| Appearance | White to off-white powder |
| Purity | ≥99.0% |
| Packing | 25kg cardboard drum or customized |
Industrial Applications
This intermediate plays a vital role in the synthesis of several important pharmaceutical compounds. It is primarily used as a key precursor in the manufacturing of triptan-class medications, which are widely prescribed for the acute treatment of migraine headaches. The high purity of our supplied material ensures that the subsequent synthesis steps proceed with optimal yield and minimal side reactions.
Specific applications include its use as an intermediate in the production of Sumatriptan succinate and Zolmitriptan. These end products are essential components in modern neurology and pain management therapeutics. By providing a reliable source of this intermediate, we support pharmaceutical companies in maintaining consistent production schedules and meeting regulatory compliance requirements for drug manufacturing.
Quality Assurance and Storage
Quality assurance is paramount in our operations. Each batch undergoes rigorous testing for identity, assay, and impurities before release. We maintain full traceability from raw material sourcing to final packaging. Proper storage is essential to maintain the integrity of the product. It should be stored in a cool, dry, and well-ventilated area, kept away from moisture and strong light or heat sources. Containers must remain tightly closed when not in use to prevent contamination or degradation.
We offer flexible packaging options to suit various logistical needs, including standard 25kg drums or custom configurations based on customer specifications. Our logistics team ensures safe and timely delivery globally, complying with all relevant transportation regulations for chemical goods. For technical support, bulk pricing inquiries, or sample requests, please contact our sales team directly.



