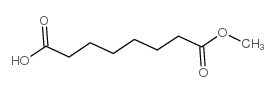
Suberic Acid Monomethyl Ester
- CAS No.3946-32-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
Suberic Acid Monomethyl Ester is a high-purity pharmaceutical intermediate essential for the synthesis of advanced therapeutics like Vorinostat. We offer bulk quantities with strict quality control and comprehensive documentation.
Request Bulk PricingProduct Technical Details
Product Overview
Suberic Acid Monomethyl Ester represents a critical component in the realm of advanced organic synthesis and pharmaceutical manufacturing. As a specialized mono-ester derivative of suberic acid, this chemical intermediate plays a pivotal role in the construction of complex molecular structures required for modern medicinal chemistry. Our facility produces this compound under stringent quality management systems to ensure consistency and reliability for downstream synthesis processes.
The compound is characterized by its distinct physical properties, including a specific density and boiling point range that facilitate efficient purification and handling during multi-step synthesis reactions. Maintaining high purity levels is paramount when utilizing this material for the production of active pharmaceutical ingredients, as impurities can significantly impact the efficacy and safety of the final therapeutic product.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 3946-32-5 |
| Molecular Formula | C9H16O4 |
| Molecular Weight | 188.22 g/mol |
| Appearance | White crystal or colorless liquid |
| Assay | ≥98.0% |
| Boiling Point | 185-186ºC at 18 mm Hg |
| Melting Point | 17-19ºC |
| Density | 1.047 g/cm3 |
| Flash Point | 113ºC |
Industrial Applications
This intermediate is primarily utilized in the pharmaceutical sector for the synthesis of histone deacetylase inhibitors and related therapeutic agents. Notably, it serves as a key building block in the manufacturing process of Vorinostat, a significant compound in oncology treatment protocols. Beyond specific drug synthesis, Suberic Acid Monomethyl Ester is valuable in research and development laboratories focusing on lipid metabolism studies and polymer chemistry applications.
Chemists and process engineers value this material for its reactivity profile, which allows for selective functionalization during esterification and amidation reactions. The stability of the compound under standard storage conditions makes it an ideal choice for large-scale production campaigns where supply chain continuity is essential.
Quality Assurance and Handling
We adhere to rigorous quality control standards to guarantee that every batch meets or exceeds the specified assay requirements. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) detailing physical constants and purity metrics. Our packaging options include standard 25 kg drums, with flexibility to accommodate custom requirements based on project scale and logistical needs.
- Consistent batch-to-batch reproducibility for reliable synthesis outcomes.
- Advanced purification techniques to minimize trace impurities.
- Secure packaging to prevent moisture absorption and contamination.
- Global shipping capabilities with compliant hazardous material documentation.
- Dedicated technical support for process optimization and troubleshooting.
Proper storage involves keeping the material in a cool, ventilated environment away from direct sunlight and incompatible substances. By following recommended handling procedures, users can maintain the integrity of the chemical throughout its shelf life. Our commitment to safety and quality ensures that partners receive a product that supports their regulatory compliance and manufacturing excellence goals.



