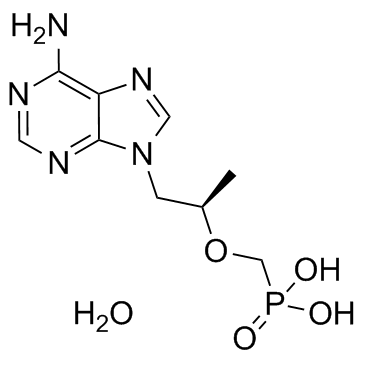
Tenofovir Hydrate
- CAS No.206184-49-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Tenofovir Hydrate (CAS 206184-49-8) designed for pharmaceutical synthesis and research applications.
Request Bulk PricingProduct Technical Details
Product Overview
Tenofovir Hydrate represents a critical nucleotide analog intermediate within the modern pharmaceutical landscape. Specifically identified as 9-[(R)-2-(Phosphonomethoxy)propyl]adenine monohydrate, this compound serves as a foundational building block for the synthesis of advanced antiviral therapies. Our facility specializes in the production of high-grade chemical intermediates that meet stringent international quality standards. This product is primarily utilized in the manufacturing pipeline for esterified derivatives, playing an indispensable role in the development of therapeutic agents targeting viral replication mechanisms.
As a dedicated B2B chemical manufacturer, we understand the importance of consistency and reliability in the supply chain. Our Tenofovir Hydrate is produced under controlled conditions to ensure minimal variability between batches. This stability is crucial for downstream processors who require predictable reaction outcomes during multi-step synthesis protocols. We commit to providing comprehensive technical documentation to support your regulatory filings and quality assurance processes.
Technical Specifications
Quality control is paramount in pharmaceutical intermediate manufacturing. Each batch undergoes rigorous analytical testing to verify compliance with specified parameters. The following table outlines the key physical and chemical properties guaranteed for this product line.
| Parameter | Specification |
|---|---|
| CAS Number | 206184-49-8 |
| Molecular Formula | C9H16N5O5P |
| Molecular Weight | 305.23 g/mol |
| Appearance | White Powder |
| Assay (Purity) | ≥98.0% |
| Loss on Drying | ≤2.0% |
| Heavy Metals | ≤10 ppm |
| Water Content | ≤1.0% |
| Sulphated Ash | ≤0.5% |
| Total Impurities | ≤0.5% |
Industrial Applications
This chemical intermediate is extensively utilized in the pharmaceutical sector for the synthesis of prodrug forms. Specifically, it acts as a key precursor in the production of Tenofovir Disoproxil Fumarate. The high purity level ensures that subsequent esterification reactions proceed with optimal yield and minimal side-product formation. Researchers and process chemists value this material for its consistent reactivity profile, which facilitates scalable manufacturing processes.
Beyond direct synthesis, this compound is also employed in analytical research and method development laboratories. Its well-defined structural characteristics make it an ideal reference standard for chromatography and spectroscopy calibration. We support various scales of operation, from early-stage research quantities to large-scale commercial production requirements.
Quality Assurance and Safety
Our manufacturing processes adhere to strict quality management systems. We provide a Certificate of Analysis (COA) with every shipment, detailing the specific test results for the batch provided. This transparency allows our partners to maintain their own quality records with confidence. Safety data sheets (SDS) are available upon request to ensure safe handling and storage within your facility.
- Consistent batch-to-batch reproducibility
- Comprehensive impurity profiling
- Secure packaging to prevent moisture uptake
- Global logistics support for timely delivery
Storage and Handling
To maintain product integrity, Tenofovir Hydrate should be stored in a cool, dry, and well-ventilated area. Containers must remain tightly closed when not in use to protect against moisture and strong light. Exposure to excessive heat should be avoided to prevent degradation. Proper personal protective equipment is recommended during handling to ensure operator safety. We recommend consulting the provided safety data sheet for detailed handling instructions and emergency measures.



