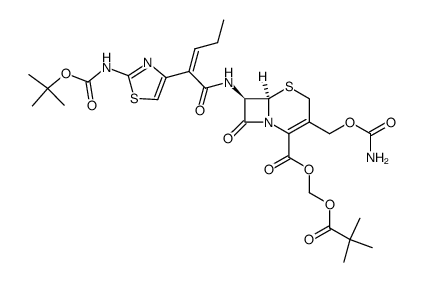
(tert-Butoxycarbonyl)oxycefcapene pivoxil
- CAS No.105889-80-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate designed for the efficient synthesis of cefcapene pivoxil hydrochloride. Reliable supply with comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
(tert-Butoxycarbonyl)oxycefcapene pivoxil is a specialized pharmaceutical intermediate critical for the production of advanced cephalosporin antibiotics. This compound serves as a key precursor in the manufacturing of cefcapene pivoxil hydrochloride, a widely recognized oral antibiotic used in clinical settings. Our facility ensures that every batch meets stringent quality standards required for pharmaceutical synthesis, providing a stable and reliable supply chain for global manufacturers.
The chemical structure incorporates a tert-butoxycarbonyl protecting group, which enhances stability during the synthesis process while ensuring high reactivity for downstream transformations. This balance is essential for maintaining the integrity of the beta-lactam ring, a crucial feature for the biological activity of the final antibiotic product. Our production processes are optimized to minimize impurities, ensuring that the intermediate supports high-yield final synthesis.
Key Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | (tert-Butoxycarbonyl)oxycefcapene pivoxil |
| CAS Number | 105889-80-3 |
| Molecular Formula | C28H37N5O10S2 |
| Molecular Weight | 667.75 g/mol |
| Appearance | Off-white to white solid |
| Purity | ≥99.0% |
| Packing | 25 kg/drum or customized |
Industrial Applications
This intermediate is primarily utilized in the pharmaceutical industry for the synthesis of cefcapene pivoxil hydrochloride. The high purity level ensures that the subsequent chemical reactions proceed efficiently, reducing waste and improving the overall yield of the final active pharmaceutical ingredient (API). It is suitable for large-scale manufacturing processes where consistency and quality are paramount.
Manufacturers relying on this intermediate benefit from its robust performance in organic synthesis routes. The compound is designed to integrate seamlessly into existing production lines, offering a drop-in solution for facilities producing cephalosporin-based antibiotics. Our technical team supports clients with synthesis route optimization to maximize efficiency.
Quality Assurance and Storage
Quality control is integral to our manufacturing process. Each batch undergoes rigorous testing using advanced analytical techniques such as HPLC and NMR to verify identity and purity. A Certificate of Analysis (COA) is provided with every shipment, documenting compliance with specified parameters. We adhere to strict Good Manufacturing Practices (GMP) guidelines to ensure product safety and consistency.
For optimal stability, the product should be stored in a cool, ventilated place away from direct sunlight and moisture. Proper storage conditions maintain the chemical integrity of the tert-butoxycarbonyl group, preventing premature degradation. We offer flexible packaging options, including 25 kg drums, to meet diverse logistical requirements. Global shipping is available, ensuring timely delivery to manufacturing sites worldwide.



