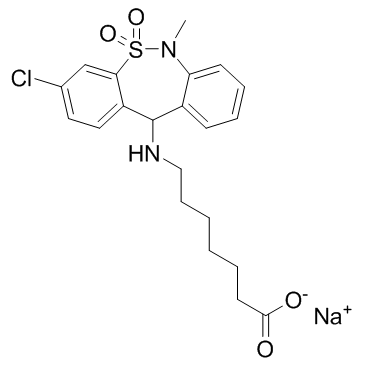
Tianeptine Sodium Salt Hydrate
- CAS No.30123-17-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Tianeptine Sodium Salt Hydrate for pharmaceutical research and synthesis applications.
Request Bulk PricingProduct Technical Details
Product Overview
Tianeptine Sodium Salt Hydrate is a specialized pharmaceutical intermediate characterized by its high chemical stability and purity. This compound serves as a critical building block in the development of advanced neurological therapeutics and biochemical research projects. Our manufacturing process ensures that each batch meets rigorous international standards for pharmaceutical intermediates, providing researchers and industrial partners with reliable material for synthesis and analysis.
As a sodium salt formulation, this substance offers improved solubility profiles compared to its free acid counterparts, making it particularly suitable for various laboratory applications and formulation studies. The hydrate form ensures stable crystalline structure under standard storage conditions, facilitating long-term inventory management for large-scale production facilities.
Chemical Specifications
Quality control is paramount in the production of fine chemical intermediates. We employ advanced analytical techniques including HPLC, NMR, and Mass Spectrometry to verify the identity and purity of every lot. The following table outlines the key physical and chemical parameters associated with this product.
| Parameter | Specification |
|---|---|
| CAS Number | 30123-17-2 |
| Molecular Formula | C21H24ClN2NaO4S |
| Molecular Weight | 458.934 g/mol |
| Appearance | White powder |
| Assay | ≥99.0% |
| Water Content | ≤0.50% |
| Density | 1.38 g/cm3 |
| Storage Condition | Desiccate at RT |
Industrial Applications
This chemical intermediate is primarily utilized in the pharmaceutical sector for the synthesis of complex organic molecules. It is frequently referenced in literature regarding neurological research and receptor binding studies. Due to its specific structural configuration involving the dibenzothiazepine core, it serves as a valuable reference standard and synthesis precursor for developing new therapeutic agents.
Industrial clients utilize this material for process development, scale-up studies, and quality assurance benchmarking. The consistent purity profile allows for reproducible results in downstream processing, reducing variability in final product formulations. Our supply chain is optimized to support both small-scale research requirements and large-volume manufacturing needs.
Storage and Handling
Proper storage is essential to maintain the integrity of Tianeptine Sodium Salt Hydrate. The material should be kept in a cool, dry place away from direct sunlight and moisture. Containers must remain tightly closed when not in use to prevent hydration changes or contamination. We recommend storing the product in its original packaging under desiccated conditions at room temperature.
Safety handling procedures should follow standard laboratory protocols for chemical intermediates. Personnel should wear appropriate personal protective equipment including gloves and safety goggles during handling. In case of spillage, standard chemical cleanup procedures should be implemented immediately to ensure workplace safety and environmental compliance.
Packaging and Logistics
We offer flexible packaging solutions to meet diverse customer requirements. Standard packaging includes 25kg drums designed for secure international shipping. Custom packaging configurations are available upon request to accommodate specific logistical needs or production line requirements. All shipments are accompanied by a Certificate of Analysis (COA) and relevant safety documentation.



