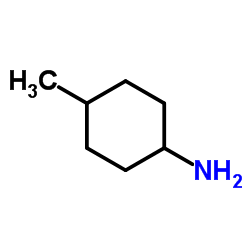
trans-4-Methylcyclohexyl amine
- CAS No.2523-55-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity trans-4-Methylcyclohexyl amine (CAS 2523-55-9) designed for pharmaceutical synthesis. Reliable supply for Glimepiride intermediate production with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
trans-4-Methylcyclohexyl amine is a specialized organic compound widely recognized for its critical role in pharmaceutical manufacturing. As a key structural component, this amine derivative serves as an essential building block in the synthesis of advanced therapeutic agents. Our facility produces this chemical under rigorous quality assurance protocols to ensure consistency and reliability for downstream applications.
The compound is characterized by its stable cyclohexane ring structure with a trans-configured methyl and amino group. This specific stereochemistry is vital for achieving the desired biological activity in final drug products. We supply this material in bulk quantities to support large-scale industrial production needs.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 2523-55-9 |
| Molecular Formula | C7H15N |
| Molecular Weight | 113.201 g/mol |
| Appearance | Colorless to yellow transparent liquid |
| Assay (Purity) | ≥99.0% |
| Density | 0.8±0.1 g/cm3 |
| Boiling Point | 149.3±8.0 °C at 760 mmHg |
| Flash Point | 26.7±0.0 °C |
| Refractive Index | 1.449 |
Industrial Applications
The primary application of trans-4-Methylcyclohexyl amine lies within the pharmaceutical sector, specifically as an intermediate in the production of antidiabetic medications. It is a crucial precursor in the synthesis pathway of Glimepiride, a widely prescribed sulfonylurea used for managing type 2 diabetes. The high purity of our supplied material ensures optimal reaction yields and minimizes impurity profiles in the final active pharmaceutical ingredient.
Beyond pharmaceuticals, this amine finds utility in fine chemical synthesis where specific steric hindrance and nucleophilic properties are required. Researchers and process chemists utilize this building block for developing novel organic compounds and optimizing synthetic routes.
Quality Assurance and Safety
We adhere to strict international standards for chemical manufacturing. Every batch undergoes comprehensive testing using advanced analytical techniques such as GC and HPLC to verify purity and identity. A Certificate of Analysis (COA) is provided with every shipment to document compliance with specifications.
- Consistent batch-to-batch quality for reliable production scaling.
- Packaged in secure drums to prevent contamination during transit.
- Full regulatory documentation support for global export.
Regarding safety, this material should be handled in a well-ventilated environment due to its flash point and vapor pressure. Proper personal protective equipment (PPE) is recommended during handling and storage. We advise storing the product in a cool, ventilated place away from incompatible substances to maintain stability.
Supply and Logistics
As a global manufacturer, we offer flexible packaging solutions including standard 200 kg drums or custom configurations based on client requirements. Our logistics network ensures timely delivery to major pharmaceutical hubs worldwide. For technical inquiries or bulk pricing requests, our sales team is available to provide detailed support and samples upon qualification.



