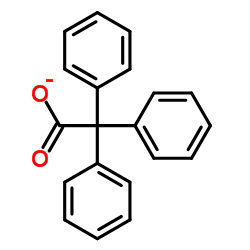
Triphenylacetic Acid
- CAS No.595-91-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity triphenylacetic acid (98%) used as a salt-forming excipient in pharmaceutical development and organic synthesis.
Request Bulk PricingProduct Technical Details
Product Overview
Triphenylacetic acid (CAS 595-91-5) is a specialized aromatic carboxylic acid widely employed in the pharmaceutical industry as a salt-forming excipient. With a molecular formula of C20H16O2 and a molecular weight of 288.34 g/mol, this compound exhibits high chemical stability under standard storage conditions and is valued for its role in modulating the physicochemical properties of active pharmaceutical ingredients (APIs), including vilanterol and related therapeutics.
Specifications
| Molecular Formula | C20H16O2 |
|---|---|
| Molecular Weight | 288.34 g/mol |
| Purity | ≥98% |
| Appearance | White to beige fine crystalline powder |
| Melting Point | 270–273 °C (lit.) |
| Boiling Point | 401.4 °C at 760 mmHg |
| Flash Point | 210.7 °C |
| Stability | Stable under normal temperatures and pressures |
| Storage Conditions | Store in a cool, dry place. Keep container tightly closed when not in use. |
Industrial Applications
As a key intermediate in advanced organic synthesis, triphenylacetic acid serves primarily as a counterion in the formation of stable, crystalline salts with basic drug molecules. This functionality enhances solubility, bioavailability, and shelf-life of final dosage forms. Its robust aromatic structure provides steric bulk that can influence crystal packing and polymorphism—critical factors in solid-state pharmaceutical development.
- Used as a salt-forming agent for bronchodilators and other respiratory therapeutics
- Serves as a building block in the synthesis of complex organic molecules
- Compatible with standard API purification and isolation workflows
- Meets stringent quality requirements for use in GMP-compliant manufacturing
Our triphenylacetic acid is manufactured under rigorous quality control protocols, ensuring batch-to-batch consistency and compliance with international regulatory standards for pharmaceutical intermediates.



