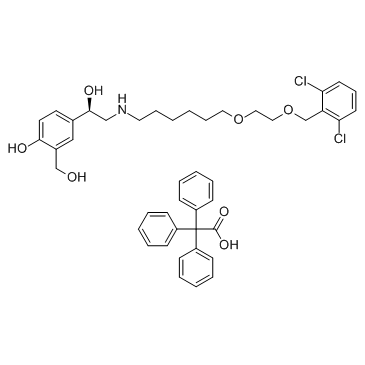
Vilanterol Trifenate
- CAS No.503070-58-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Vilanterol Trifenate intermediate for respiratory pharmaceutical development and research purposes.
Request Bulk PricingProduct Technical Details
Product Overview
Vilanterol Trifenate is a high-value pharmaceutical intermediate characterized by its specific chemical structure designed for respiratory therapeutic applications. As a salt form of the long-acting beta2-adrenergic agonist Vilanterol, this compound plays a critical role in the synthesis and formulation of advanced inhalation therapies. Our supply chain ensures that every batch meets rigorous international standards for purity and consistency, making it an ideal choice for pharmaceutical research and development teams focused on respiratory health solutions.
This chemical entity is meticulously processed to maintain stability and potency. It serves as a key building block in the production of medications aimed at managing chronic obstructive pulmonary disease and asthma. By providing a reliable source of this intermediate, we support the global pharmaceutical industry in delivering effective treatments to patients worldwide.
Chemical Specifications
The following table outlines the critical technical parameters associated with this product. Our quality control team verifies each parameter through advanced analytical methods including HPLC and NMR spectroscopy.
| Parameter | Specification |
|---|---|
| CAS Number | 503070-58-4 |
| Molecular Formula | C44H49Cl2NO7 |
| Molecular Weight | 774.77 g/mol |
| Purity | NLT 98% |
| Appearance | White Powder |
| Storage Conditions | 2-8°C, Protected from Light |
Quality Assurance
We understand that consistency is paramount in pharmaceutical manufacturing. Our facility operates under strict quality management systems to ensure that Vilanterol Trifenate is produced with minimal batch-to-batch variation. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) that details the results of all relevant tests. This documentation supports regulatory filings and ensures transparency throughout the supply chain.
Our commitment to quality extends beyond mere compliance. We employ state-of-the-art purification techniques to remove impurities and residual solvents. This dedication to excellence ensures that our clients receive a product that is ready for immediate use in sensitive formulation processes without the need for additional purification steps.
Industrial Applications
Primarily utilized in the pharmaceutical sector, this intermediate is essential for the development of respiratory medications. It is compatible with various dry powder inhaler technologies and nebulizer formulations. Research teams use this material to establish performance benchmarks and validate new delivery systems. The high purity profile allows for accurate dosing and reliable efficacy data during preclinical and clinical trials.
Additionally, this compound serves as a reference standard for analytical laboratories conducting quality control on finished drug products. Its stability profile makes it suitable for long-term studies regarding drug degradation and shelf-life estimation. By choosing our supply, manufacturers gain a partner dedicated to supporting the entire lifecycle of respiratory therapeutic development.
Storage and Handling
To maintain the integrity of Vilanterol Trifenate, proper storage protocols must be followed. The material should be kept in a cool, dry environment away from direct sunlight and moisture sources. Containers should remain sealed when not in use to prevent hygroscopic absorption. Handling should be conducted in a well-ventilated area with appropriate personal protective equipment to ensure safety during weighing and transfer operations.



