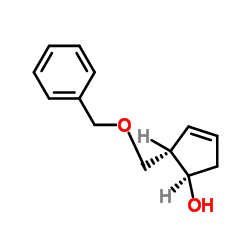
(1R,2S)-2-(phenylmethoxymethyl)cyclopent-3-en-1-ol
- CAS No.188399-48-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate crucial for Entecavir synthesis. Available in bulk with comprehensive COA and global shipping options.
Request Bulk PricingProduct Technical Details
Product Overview
(1R,2S)-2-(phenylmethoxymethyl)cyclopent-3-en-1-ol is a specialized chiral intermediate designed for the pharmaceutical industry. This compound serves as a critical building block in the multi-step synthesis of Entecavir, a potent antiviral agent used in the treatment of chronic hepatitis B virus infections. Our manufacturing process ensures exceptional stereochemical purity, which is vital for the efficacy and safety of the final active pharmaceutical ingredient.
As a dedicated supplier of advanced organic synthesis materials, we understand the stringent requirements of modern drug development. This intermediate is produced under strictly controlled conditions to minimize impurities and maximize yield. The unique structural configuration, featuring a cyclopentene ring with specific stereocenters, requires precise synthetic routes that our facility has optimized over years of technical refinement.
Specifications and Physical Properties
Quality consistency is the cornerstone of our production philosophy. Each batch undergoes rigorous analytical testing to confirm identity and purity levels. The physical characteristics are tightly monitored to ensure compatibility with downstream processing conditions.
| Parameter | Specification |
|---|---|
| CAS Number | 188399-48-6 |
| Molecular Formula | C13H16O2 |
| Molecular Weight | 204.265 g/mol |
| Appearance | White crystalline powder |
| Assay (Purity) | ≥98.0% |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 318.9±22.0 °C at 760 mmHg |
| Flash Point | 135.5±16.6 °C |
Industrial Applications
The primary application of this chemical is as a key intermediate in the manufacturing of Entecavir. The stereochemistry of the molecule must be preserved throughout the synthesis pathway to ensure the biological activity of the final drug product. Our material is suitable for large-scale production environments where reproducibility and reliability are paramount.
- Essential precursor for antiviral pharmaceutical synthesis
- Compatible with standard organic transformation protocols
- Supports GMP-compliant manufacturing workflows
- Optimized for high-yield coupling reactions
Quality Assurance and Packaging
We provide a comprehensive Certificate of Analysis (COA) with every shipment, detailing all relevant test results including HPLC purity, residual solvents, and heavy metals. Our quality control team adheres to international standards to guarantee that every drum meets the specified criteria. Packaging is designed to maintain stability during transit and storage.
Standard packaging consists of 25 kg drums, though we offer flexible solutions to meet specific customer requirements. Proper storage in a cool, ventilated area is recommended to preserve the integrity of the compound. Our logistics network ensures timely global delivery, supporting your production schedules without interruption. For technical inquiries or custom synthesis requests, our expert team is available to provide detailed support.



