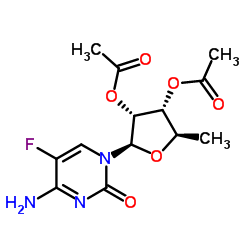
2',3'-di-O-acetyl-5'-deoxy-5-fluorocytidine
- CAS No.161599-46-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
Premium grade pharmaceutical intermediate for Capecitabine synthesis with high purity assurance and global supply capabilities.
Request Bulk PricingProduct Technical Details
Product Overview
2',3'-di-O-acetyl-5'-deoxy-5-fluorocytidine is a highly specialized pharmaceutical intermediate essential for the synthesis of advanced anticancer therapeutics. As a key precursor in the manufacturing pathway of Capecitabine, this compound plays a critical role in modern oncology drug production. Our facility produces this nucleoside analogue under strict quality control protocols to ensure consistent molecular integrity and high chemical purity suitable for large-scale pharmaceutical applications.
We understand the stringent requirements of the global pharmaceutical supply chain. Therefore, our production processes are designed to meet international standards, providing clients with reliable bulk quantities that support continuous manufacturing operations. This product is specifically engineered for use in professional laboratory and industrial synthesis environments.
Chemical Properties
The following physical and chemical data characterizes the standard profile of this intermediate. These properties are critical for process engineers when designing synthesis routes and handling protocols.
| Property | Value |
|---|---|
| CAS Number | 161599-46-8 |
| Molecular Formula | C13H16FN3O6 |
| Molecular Weight | 329.281 g/mol |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 427.4±55.0 °C at 760 mmHg |
| Melting Point | 196-198 °C |
| Flash Point | 212.3±31.5 °C |
Quality Specifications
Quality assurance is paramount in pharmaceutical intermediate manufacturing. We employ advanced analytical techniques including HPLC and NMR to verify that every batch meets or exceeds the specified purity thresholds. Our commitment to quality ensures downstream synthesis efficiency and final drug safety.
| Parameter | Specification |
|---|---|
| Appearance | White to light yellow powder |
| Assay (Purity) | ≥98.0% |
| Moisture Content | ≤1.0% |
| Residue on Ignition | ≤1.0% |
| Single Impurity | ≤0.3% |
| Total Impurities | ≤2.0% |
Industrial Applications
This compound is primarily utilized as a building block in the multi-step synthesis of Capecitabine, an oral chemotherapy medication. Its specific structural configuration allows for precise chemical transformations required in nucleoside analogue production. Key advantages of sourcing from our facility include:
- Consistent batch-to-batch reproducibility for stable manufacturing processes.
- High purity levels that minimize downstream purification costs.
- Scalable production capacity to meet fluctuating market demands.
- Comprehensive documentation including Certificates of Analysis (COA).
Storage and Handling
To maintain chemical stability and safety, proper storage conditions must be observed. The product should be kept in a cool, ventilated area away from direct sunlight and moisture sources. Containers must remain tightly sealed when not in use to prevent degradation. Personnel handling this material should adhere to standard laboratory safety practices and wear appropriate personal protective equipment. We offer flexible packaging options, including 25 kg drums, and can customize packaging solutions based on specific logistical requirements.



