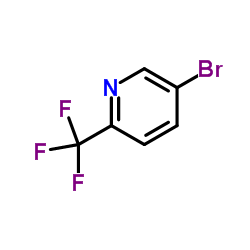
2-Trifluoromethyl-5-bromopyridine
- CAS No.436799-32-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 2-Trifluoromethyl-5-bromopyridine (CAS 436799-32-5) designed for pharmaceutical synthesis. Reliable supply with strict quality control for global research and production needs.
Request Bulk PricingProduct Technical Details
Product Overview
2-Trifluoromethyl-5-bromopyridine is a highly specialized fluorinated heterocyclic compound that serves as a critical building block in modern medicinal chemistry and organic synthesis. As a key intermediate, this substance features a pyridine ring substituted with both a bromine atom and a trifluoromethyl group, providing unique reactivity profiles essential for complex molecule construction. The incorporation of the trifluoromethyl moiety is particularly valued in drug design due to its ability to enhance metabolic stability, lipophilicity, and bioavailability of final pharmaceutical agents.
Our manufacturing process ensures that every batch meets rigorous international standards for purity and consistency. This compound is extensively utilized by research laboratories and industrial production facilities focused on developing advanced therapeutic solutions. The strategic positioning of the bromine atom allows for versatile cross-coupling reactions, making it an indispensable tool for chemists aiming to construct diverse chemical libraries.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 436799-32-5 |
| Chemical Name | 2-Trifluoromethyl-5-bromopyridine |
| Molecular Formula | C6H3BrF3N |
| Molecular Weight | 225.99 g/mol |
| Purity | ≥98.0% |
| Appearance | Crystal powder |
| Melting Point | 44-46 °C |
| Boiling Point | 179.1±35.0 °C at 760 mmHg |
| Density | 1.7±0.1 g/cm3 |
| Refractive Index | 1.471 |
Industrial Applications
This fluorinated pyridine derivative is primarily employed as a pharmaceutical intermediate in the synthesis of active pharmaceutical ingredients (APIs). Its structural characteristics enable participation in various catalytic transformations, including Suzuki-Miyaura coupling, nucleophilic aromatic substitution, and metal-halogen exchange reactions. The presence of the electron-withdrawing trifluoromethyl group significantly influences the electronic properties of the pyridine ring, facilitating specific reaction pathways that are difficult to achieve with non-fluorinated analogs.
- Essential building block for kinase inhibitors and receptor modulators.
- Used in the development of agrochemicals and fine chemicals.
- Supports high-throughput screening campaigns in drug discovery.
- Compatible with diverse synthetic routes for scale-up production.
Quality Assurance and Storage
We maintain a comprehensive quality management system to guarantee the integrity of 2-Trifluoromethyl-5-bromopyridine from synthesis to delivery. Each production lot undergoes strict analytical testing using advanced chromatography and spectroscopy methods to verify identity and purity levels. A Certificate of Analysis (COA) is provided with every shipment to ensure full traceability and compliance with customer specifications.
For optimal stability, the product should be stored in a tightly closed container within a cool, dry, and well-ventilated area. Protection from moisture and direct sunlight is recommended to maintain physical and chemical properties over time. Standard packaging involves 25 kg drums, though custom packaging solutions are available upon request to accommodate specific logistical requirements for global shipping.



