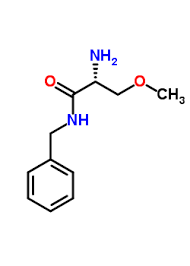
(2R)-2-amino-N-benzyl-3-methoxypropanamide
- CAS No.196601-69-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity (2R)-2-amino-N-benzyl-3-methoxypropanamide serves as a critical chiral intermediate for Lacosamide synthesis. We provide reliable bulk supply with full quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
(2R)-2-amino-N-benzyl-3-methoxypropanamide is a specialized chiral pharmaceutical intermediate designed for the synthesis of advanced anticonvulsant agents. This compound features a specific stereochemical configuration essential for maintaining the biological activity of downstream therapeutic products. Our manufacturing process ensures exceptional optical purity and chemical consistency, meeting the rigorous demands of modern pharmaceutical development.
As a key building block in the production of Lacosamide, this amide derivative plays a pivotal role in establishing the molecular framework required for effective neurological therapeutics. We prioritize strict quality control measures at every stage of production to guarantee compliance with international regulatory standards.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 196601-69-1 |
| Molecular Formula | C11H16N2O2 |
| Molecular Weight | 208.26 g/mol |
| Appearance | White to off-white powder |
| Assay | ≥98.0% |
| Density | 1.107 g/cm3 |
| Boiling Point | 421.95°C at 760 mmHg |
| Flash Point | 208.99°C |
Industrial Applications
This intermediate is primarily utilized in the pharmaceutical industry for the synthesis of Lacosamide, a widely recognized antiepileptic drug. The high enantiomeric excess provided by our product ensures optimal reaction yields and minimizes the formation of unwanted isomers during the manufacturing process. It is suitable for large-scale commercial production as well as research and development initiatives focused on neurological disorders.
- Essential precursor for anticonvulsant drug synthesis
- Supports GMP-compliant manufacturing workflows
- Compatible with various organic synthesis routes
- Ideal for custom pharmaceutical development projects
Quality Assurance and Storage
Every batch undergoes comprehensive analytical testing, including HPLC, NMR, and mass spectrometry, to verify identity and purity. We provide a Certificate of Analysis (COA) with each shipment to ensure transparency and traceability. To maintain stability, the product should be stored in a cool, ventilated environment away from direct sunlight and moisture. Proper handling procedures must be followed to preserve chemical integrity during transportation and usage.
Packaging and Logistics
We offer flexible packaging solutions to meet diverse client needs, typically supplying in 25 kg drums. Custom packaging configurations are available upon request. Our global logistics network ensures timely delivery while maintaining product safety throughout the supply chain. Partner with us for reliable access to high-quality pharmaceutical intermediates backed by decades of industry expertise.



