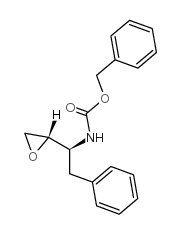
(2S,3S)-1,2-Epoxy-3-(Cbz-amino)-4-phenylbutane
- CAS No.128018-44-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity chiral intermediate essential for the synthesis of Saquinavir. Manufactured with strict stereochemical control and quality assurance for global pharmaceutical supply chains.
Request Bulk PricingProduct Technical Details
Product Overview
(2S,3S)-1,2-Epoxy-3-(Cbz-amino)-4-phenylbutane is a highly specialized chiral building block utilized extensively in the pharmaceutical industry. This compound serves as a critical intermediate in the total synthesis of Saquinavir, a prominent protease inhibitor used in antiretroviral therapy. The molecule features a strategically protected amino group via the benzyloxycarbonyl (Cbz) moiety and a reactive epoxide ring, enabling precise downstream chemical transformations. Our manufacturing process ensures exceptional stereochemical integrity, specifically maintaining the (2S,3S) configuration which is vital for the biological activity of the final API.
Key Specifications
| Parameter | Value |
|---|---|
| CAS Number | 128018-44-0 |
| Molecular Formula | C18H19NO3 |
| Molecular Weight | 297.35 g/mol |
| Appearance | White to light yellow powder |
| Assay (Purity) | ≥98.0% |
| Density | 1.205 g/cm3 |
| Boiling Point | 481.8°C at 760 mmHg |
Industrial Applications
Primarily employed as a pharmaceutical intermediate, this epoxide derivative is indispensable for researchers and manufacturers developing HIV protease inhibitors. The presence of the chiral epoxide allows for regioselective ring-opening reactions, facilitating the construction of complex peptide mimics. Beyond Saquinavir synthesis, this building block is valuable in medicinal chemistry programs focused on peptidomimetics and enzyme inhibitors. The Cbz protecting group offers stability during various synthetic steps while remaining removable under standard hydrogenolysis conditions, providing flexibility in multi-step synthesis routes.
Quality Control and Assurance
Quality is paramount in the production of chiral intermediates. Our facility employs advanced analytical techniques including High-Performance Liquid Chromatography (HPLC) and Nuclear Magnetic Resonance (NMR) spectroscopy to verify stereochemical purity and chemical identity. Each batch undergoes rigorous testing to ensure compliance with international pharmaceutical standards. We provide a comprehensive Certificate of Analysis (COA) with every shipment, detailing impurity profiles and confirmation of the specific (2S,3S) stereoisomer. Our quality management system is designed to meet the demanding requirements of global regulatory bodies.
Packaging and Storage
To maintain stability and prevent degradation, the product is packaged in secure, moisture-resistant containers. Standard packaging includes 25 kg drums, though we offer flexible customization to meet specific client logistics requirements. Storage recommendations include keeping the material in a cool, ventilated area away from direct sunlight and strong oxidizing agents. Proper handling procedures should be followed to ensure safety and preserve the chemical integrity of the epoxide functionality during transport and warehousing. Our global supply chain ensures timely delivery to research centers and manufacturing plants worldwide.



