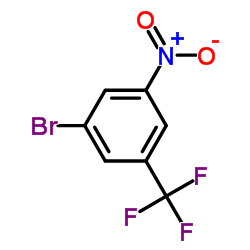
3-Bromo-5-Nitrobenzotrifluoride
- CAS No.630125-49-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 3-Bromo-5-Nitrobenzotrifluoride liquid for advanced organic synthesis. Reliable supply with ≥98.0% assay and global shipping.
Request Bulk PricingProduct Technical Details
Product Overview
3-Bromo-5-Nitrobenzotrifluoride is a specialized fluorinated aromatic compound designed for complex organic synthesis applications. As a key building block in modern medicinal chemistry, this substance features a unique substitution pattern that facilitates diverse chemical transformations. The presence of the trifluoromethyl group enhances lipophilicity and metabolic stability in downstream pharmaceutical candidates, making it a valuable asset for research and development teams.
Our manufacturing process ensures consistent quality and high purity levels suitable for sensitive synthetic routes. This intermediate is widely utilized in the preparation of agrochemicals, pharmaceuticals, and advanced materials. We adhere to strict quality control protocols to guarantee that every batch meets rigorous international standards for chemical intermediates.
Technical Specifications
The following table outlines the critical physical and chemical properties of this product. These parameters are verified through advanced analytical methods including GC-MS and NMR spectroscopy.
| Parameter | Value |
|---|---|
| Chemical Name | 3-Bromo-5-Nitrobenzotrifluoride |
| CAS Number | 630125-49-4 |
| Molecular Formula | C7H3BrF3NO2 |
| Molecular Weight | 270.00 g/mol |
| Appearance | Light Yellow Liquid |
| Purity | ≥98.0% |
| Density | 1.788 g/cm3 |
| Boiling Point | 223.7ºC at 760 mmHg |
| Flash Point | 89.1ºC |
| Refractive Index | 1.515 |
Industrial Applications
This fluorinated intermediate serves as a versatile precursor in multi-step synthesis pathways. The bromo and nitro functional groups allow for various substitution reactions, including nucleophilic aromatic substitution and palladium-catalyzed cross-coupling reactions such as Suzuki or Buchwald-Hartwig couplings. Chemists utilize this compound to introduce trifluoromethyl motifs into complex molecular scaffolds.
Common applications include the synthesis of kinase inhibitors, receptor modulators, and crop protection agents. The stability of the nitro group under specific conditions enables selective reduction to aniline derivatives, further expanding its utility in creating diverse chemical libraries. Our clients rely on this material for scaling up processes from laboratory discovery to pilot production.
Quality Assurance and Storage
We maintain a robust quality management system to ensure product integrity. Each shipment is accompanied by a Certificate of Analysis (COA) detailing batch-specific test results. Packaging is designed to prevent contamination and degradation during transit. Standard options include 1 kg bottles for laboratory use and 25 kg drums for industrial-scale operations.
For optimal stability, store this chemical in a tightly closed container within a cool, dry, and well-ventilated area. Avoid exposure to direct sunlight and strong oxidizing agents. Proper handling procedures should be followed according to safety data sheets to ensure workplace safety. Contact our technical support team for detailed storage guidelines and bulk pricing information tailored to your project requirements.



