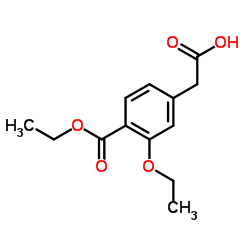
3-Ethoxy-4-(ethoxycarbonyl)phenylacetic Acid
- CAS No.99469-99-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity pharmaceutical intermediate essential for Repaglinide synthesis. Manufactured with strict quality control for global bulk supply.
Request Bulk PricingProduct Technical Details
Product Overview
3-Ethoxy-4-(ethoxycarbonyl)phenylacetic Acid is a highly specialized organic compound designed for advanced pharmaceutical synthesis. This critical intermediate plays a pivotal role in the production of Repaglinide, a well-known therapeutic agent used in the management of diabetes. Our facility produces this chemical with a focus on exceptional purity and consistency, ensuring it meets the rigorous demands of modern drug manufacturing processes.
As a key building block in the pharmaceutical industry, this acid derivative requires precise structural integrity to facilitate downstream reactions effectively. We utilize state-of-the-art synthesis routes to minimize impurities and maximize yield. Our commitment to quality ensures that every batch conforms to strict international standards, making it a reliable choice for large-scale production environments.
Key Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 99469-99-5 |
| Molecular Formula | C13H16O5 |
| Molecular Weight | 252.26 g/mol |
| Appearance | White to off-white crystalline powder |
| Purity | ≥99% |
| Melting Point | 78-80°C |
| Loss on Drying | ≤0.5% |
| Heavy Metals | ≤20ppm |
Industrial Applications
The primary application of 3-Ethoxy-4-(ethoxycarbonyl)phenylacetic Acid is as a precursor in the synthesis of Repaglinide. Its chemical structure allows for efficient coupling and functional group transformations required in the final active pharmaceutical ingredient (API) production. Beyond this specific use, it serves as a valuable component in research and development for related organic synthesis projects.
- Essential intermediate for antidiabetic medication production
- Suitable for custom pharmaceutical synthesis projects
- High stability ensures reliable processing performance
- Compatible with standard industrial reaction conditions
Quality Assurance and Storage
We maintain a robust quality management system to guarantee product safety and efficacy. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) detailing all critical parameters. To maintain stability, the product should be stored in a cool, dry, and well-closed container. It is crucial to keep the material away from moisture, strong light, and excessive heat to prevent degradation.
Our packaging options include 25kg cardboard drums or customized solutions based on client requirements. We support global logistics with secure handling procedures to ensure the product arrives in optimal condition. For bulk orders, technical support and detailed manufacturing process documentation are available upon request.



