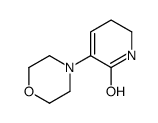
3-Morpholino-5,6-dihydropyridin-2(1H)-one
- CAS No.545445-40-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate essential for Apixaban synthesis. Available in bulk with comprehensive quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
3-Morpholino-5,6-dihydropyridin-2(1H)-one represents a critical heterocyclic building block within the modern pharmaceutical landscape. Specifically designed for high-level organic synthesis, this compound serves as a key precursor in the manufacturing of advanced cardiovascular therapeutics. Our facility specializes in the production of this intermediate with a strict focus on molecular integrity and batch-to-batch consistency.
The chemical structure incorporates a morpholine moiety fused to a dihydropyridinone ring system, providing unique reactivity profiles essential for downstream API construction. As a dedicated manufacturer, we understand that the quality of an intermediate directly impacts the efficacy and safety of the final medicinal product. Therefore, our production processes are aligned with rigorous industry standards to ensure optimal performance in complex synthesis routes.
Technical Specifications
We maintain tight control over physical and chemical parameters to meet the demanding requirements of pharmaceutical R&D and production teams. The following table outlines the standard specifications for our commercial grade material:
| Parameter | Specification |
|---|---|
| Chemical Name | 3-Morpholino-5,6-dihydropyridin-2(1H)-one |
| CAS Number | 545445-40-7 |
| Molecular Formula | C9H14N2O2 |
| Molecular Weight | 182.22 g/mol |
| Appearance | Light yellow to yellow powder |
| Assay (Purity) | ≥98.0% |
| Density | 1.195 g/cm3 |
Industrial Applications
This compound is primarily utilized as a specialized intermediate in the synthesis of Apixaban, a widely recognized anticoagulant medication. The high purity level of ≥98.0% ensures minimal impurity carryover during subsequent reaction steps, which is vital for maintaining the safety profile of the final active pharmaceutical ingredient. Beyond this specific application, the structural features of this molecule make it a valuable scaffold for medicinal chemists exploring novel therapeutic avenues involving nitrogen-containing heterocycles.
Our clients leverage this material for scale-up processes where reliability and supply chain stability are paramount. We support both pilot-scale trials and full commercial manufacturing volumes, providing the flexibility needed for dynamic project timelines.
Quality Assurance and Packaging
Quality control is embedded at every stage of our manufacturing process. Each batch undergoes comprehensive analytical testing, including HPLC and NMR verification, to confirm identity and purity. A Certificate of Analysis (COA) is provided with every shipment, ensuring full traceability and compliance with procurement requirements.
Standard packaging consists of 25 kg drums, designed to protect the material from moisture and environmental contaminants during transit. We also offer customized packaging solutions to accommodate specific logistical needs. For storage, we recommend keeping the product in a cool, ventilated area away from direct sunlight and incompatible substances to maintain stability over extended periods.
Why Choose Our Supply
- Consistent high purity levels suitable for GMP environments.
- Robust supply chain capable of meeting global demand.
- Technical support available for synthesis optimization.
- Competitive bulk pricing structures for long-term partners.
- Secure packaging ensuring product integrity upon arrival.
Partner with us for reliable access to premium pharmaceutical intermediates. Our commitment to quality and service makes us a preferred choice for innovative chemical companies worldwide.



