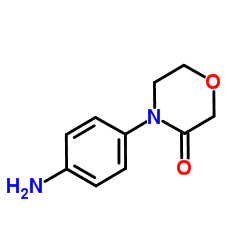
4-(4-Aminophenyl)morpholin-3-one
- CAS No.438056-69-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate crucial for anticoagulant synthesis. Manufactured under strict quality controls for global API producers.
Request Bulk PricingProduct Technical Details
Product Overview
4-(4-Aminophenyl)morpholin-3-one represents a critical structural component in the landscape of modern medicinal chemistry. As a specialized organic compound, this substance serves as a pivotal building block for the synthesis of complex pharmaceutical active ingredients. Our facility is dedicated to providing this intermediate with exceptional consistency and reliability, catering to the rigorous demands of the global pharmaceutical manufacturing sector. The compound is characterized by its stable morpholinone ring structure, which offers superior reactivity profiles suitable for multi-step synthesis pathways.
Technical Specifications
Quality and precision are paramount in the production of pharmaceutical intermediates. Each batch of 4-(4-Aminophenyl)morpholin-3-one undergoes comprehensive analytical testing to ensure it meets or exceeds industry standards. The physical and chemical properties are tightly controlled to facilitate smooth downstream processing. Below are the key specifications that define the quality of our product:
| Parameter | Specification |
|---|---|
| CAS Number | 438056-69-0 |
| Molecular Formula | C10H12N2O2 |
| Molecular Weight | 192.214 |
| Appearance | White to light yellow solid |
| Assay | ≥99.0% |
| Melting Point | 171.0 to 175.0 °C |
| Loss on Drying | ≤1.0% |
Industrial Applications
This intermediate is primarily utilized in the production of advanced anticoagulant therapies. Specifically, it acts as a key precursor in the synthesis pathway of Rivaroxaban, a widely recognized pharmaceutical agent. The high purity level of our intermediate is essential for maximizing the yield and efficiency of the final API production. Impurities at this stage can significantly impact the safety and efficacy of the downstream medication. Therefore, our manufacturing process focuses on minimizing by-products and ensuring a clean reaction profile. This makes our product an ideal choice for pharmaceutical companies seeking to optimize their synthesis routes and maintain regulatory compliance.
Quality Assurance and Manufacturing
Our production facility adheres to strict quality management systems designed to ensure product safety and consistency. We employ advanced analytical techniques, including High-Performance Liquid Chromatography (HPLC) and Nuclear Magnetic Resonance (NMR), to verify the structural integrity and purity of every lot. Our quality control team monitors critical parameters throughout the manufacturing process, from raw material sourcing to final packaging. This rigorous approach guarantees that clients receive a product that is reliable for large-scale industrial applications. We understand that supply chain stability is crucial for our partners, and we maintain robust inventory levels to meet fluctuating market demands.
Packaging and Storage
To preserve the chemical stability and integrity of 4-(4-Aminophenyl)morpholin-3-one, proper handling and storage are essential. The product is typically packaged in 25 kg drums, which are designed to protect the material from moisture and environmental contaminants. Custom packaging solutions are available upon request to accommodate specific logistical requirements. For storage, we recommend keeping the material in a cool, ventilated place away from direct sunlight and incompatible substances. Adhering to these storage guidelines ensures that the product maintains its specified properties over time. We also provide comprehensive documentation, including Certificates of Analysis (COA), with every shipment to support your regulatory filings and quality audits.



