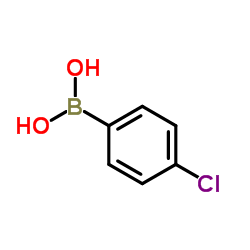
4-Chlorophenylboronic acid
- CAS No.1679-18-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 4-Chlorophenylboronic acid (CAS 1679-18-1) designed for pharmaceutical intermediate synthesis and Suzuki coupling reactions.
Request Bulk PricingProduct Technical Details
Product Overview
4-Chlorophenylboronic acid is a highly specialized organoboron compound that serves as a critical building block in modern organic synthesis. As a key aryl boronic acid derivative, this chemical is extensively utilized in the pharmaceutical and fine chemical industries for constructing complex molecular architectures. Our manufacturing process ensures exceptional batch-to-batch consistency, making it an ideal choice for large-scale production environments where reliability and purity are paramount.
This compound features a chlorophenyl group attached to a boronic acid moiety, providing unique reactivity profiles suitable for cross-coupling reactions. The stability and solubility characteristics of this material allow for versatile application in various synthetic pathways, particularly where high precision is required for downstream API development.
Technical Specifications
We adhere to strict quality control protocols to guarantee that every shipment meets or exceeds industry standards. The following table outlines the key physical and chemical properties of our premium grade product.
| Parameter | Specification |
|---|---|
| CAS Number | 1679-18-1 |
| Molecular Formula | C6H6BClO2 |
| Molecular Weight | 156.38 g/mol |
| Appearance | White to off-white powder |
| Assay (Purity) | ≥98.0% |
| Water Content | ≤0.50% |
| Melting Point | 284-289 °C |
| Density | 1.32 g/cm3 |
Industrial Applications
The primary utility of 4-Chlorophenylboronic acid lies in its role as a versatile intermediate for the synthesis of advanced pharmaceutical compounds. It is particularly valued in palladium-catalyzed cross-coupling reactions, which are fundamental to creating carbon-carbon bonds in complex organic molecules.
- Used as a key intermediate in the synthesis of oncology therapeutics such as Venetoclax and Vemurafenib.
- Essential reagent for double Suzuki coupling reactions to produce substituted diarylmethylene hydrazine derivatives.
- Employed in the development of agrochemicals and functional materials requiring precise aryl substitution.
- Utilized in medicinal chemistry research for library synthesis and structure-activity relationship studies.
Quality Assurance And Safety
Our facility operates under rigorous quality management systems to ensure product integrity. Each batch undergoes comprehensive testing using advanced analytical techniques including HPLC and NMR spectroscopy. We provide a Certificate of Analysis (COA) with every order to verify compliance with specified parameters. Regarding safety, while this product is classified as non-dangerous goods for transport, standard laboratory safety practices should be followed. Personnel should wear appropriate personal protective equipment including gloves and safety goggles during handling to prevent irritation.
Packaging And Storage
To maintain stability and prevent degradation, 4-Chlorophenylboronic acid is packaged in secure, moisture-resistant containers. Standard packaging includes 25kg drums, though custom packaging solutions are available to meet specific logistical requirements. The product should be stored in a cool, dry, and well-ventilated area. Containers must remain tightly closed when not in use to protect against moisture absorption, which could affect purity levels over time. Proper storage ensures the material retains its specified performance characteristics throughout its shelf life.



