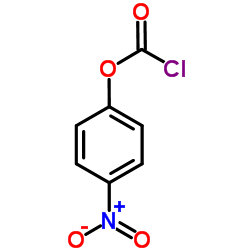
4-Nitrophenyl chloroformate
- CAS No.7693-46-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 4-Nitrophenyl chloroformate (CAS 7693-46-1) serves as a critical activating agent for organic synthesis and pharmaceutical intermediate production.
Request Bulk PricingProduct Technical Details
Product Overview
4-Nitrophenyl chloroformate is a highly reactive chemical reagent extensively utilized in organic synthesis and medicinal chemistry. Functioning primarily as an activating agent, it facilitates the conversion of hydroxyl groups into excellent leaving groups, enabling efficient nucleophilic substitution reactions. This compound is indispensable for the synthesis of carbamates, urethanes, and carbonates, which are foundational structures in numerous pharmaceutical active ingredients. Our manufacturing process ensures exceptional consistency and reliability for large-scale industrial applications.
As a key building block in the pharmaceutical industry, this chloroformate derivative supports the production of complex therapeutic agents. It is particularly valued for its ability to introduce the p-nitrophenoxycarbonyl protecting group, which can be subsequently removed under mild conditions. This versatility makes it a preferred choice for process chemists developing synthesis routes for antiretroviral and oncology pharmaceutical APIs.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 7693-46-1 |
| Molecular Formula | C7H4ClNO4 |
| Molecular Weight | 201.56 g/mol |
| Appearance | White to off-white crystalline powder |
| Purity | ≥99.0% |
| Melting Point | 77-79 °C |
| Boiling Point | 159-162 °C at 19 mm Hg |
| Water Content | ≤0.1% |
| Heavy Metals | ≤10 ppm |
| Loss on Drying | ≤0.1% |
Applications in Pharmaceutical Synthesis
The primary utility of 4-Nitrophenyl chloroformate lies in its role as a coupling agent and protecting group introducer. In the context of pharmaceutical manufacturing, it is employed to activate alcohols and amines during the construction of complex molecular architectures. High industrial purity is critical to prevent side reactions that could compromise the yield or safety of the final drug substance. Our product is rigorously tested to meet the stringent requirements of modern Good Manufacturing Practices (GMP) for intermediate supply.
Common downstream applications include the synthesis of critical antiviral and antineoplastic agents. The reagent's reactivity profile allows for precise control over reaction kinetics, ensuring high conversion rates in multi-step synthesis pathways. Whether used in laboratory-scale process development or ton-scale commercial production, consistent quality is maintained to support regulatory filings and supply chain stability.
Quality Assurance and Storage
We adhere to strict quality control protocols to guarantee the integrity of every batch. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) detailing physical and chemical properties. To maintain stability, the product must be stored in a cool, dry, and well-ventilated area away from moisture and strong light. The recommended storage temperature is between 2-8°C in tightly closed containers to prevent hydrolysis. We offer flexible packaging options, including 25kg cardboard drums, customized to meet specific logistical requirements for global distribution.



