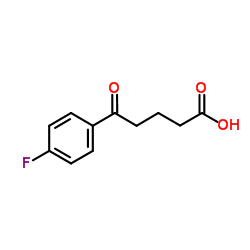
5-(4-Fluorophenyl)-5-oxopentanoic acid
- CAS No.149437-76-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used in the synthesis of Ezetimibe. Available in bulk quantities with strict quality control and comprehensive documentation.
Request Bulk PricingProduct Technical Details
Product Overview
5-(4-Fluorophenyl)-5-oxopentanoic acid is a specialized organic compound serving as a critical building block in modern medicinal chemistry. This fluorinated intermediate is primarily utilized in the pharmaceutical industry for the synthesis of lipid-lowering agents, most notably Ezetimibe. Our manufacturing process ensures consistent molecular integrity and high purity levels required for downstream pharmaceutical applications. The presence of the fluorophenyl group enhances the metabolic stability and bioavailability of the final drug product, making this intermediate essential for effective therapeutic formulations.
Technical Specifications
We adhere to rigorous quality standards to guarantee that every batch meets the specific requirements for pharmaceutical synthesis. The following table outlines the key physical and chemical parameters verified through our internal quality control laboratories.
| Parameter | Specification |
|---|---|
| CAS Number | 149437-76-3 |
| Molecular Formula | C11H11FO3 |
| Molecular Weight | 210.20 g/mol |
| Appearance | White crystal powder |
| Purity | ≥98.0% |
| Loss on Drying | ≤0.5% |
| Melting Point | 142-144 °C |
| Residual Solvents | Compliant with ICH Guidelines |
Industrial Applications
The primary application of 5-(4-Fluorophenyl)-5-oxopentanoic acid lies in the production of cholesterol absorption inhibitors. It acts as a key precursor in the multi-step synthesis route of Ezetimibe, facilitating the construction of the core molecular structure. Beyond this specific use, the compound serves as a valuable reagent in organic synthesis laboratories for developing novel fluorinated analogs. Its chemical stability and reactivity profile make it suitable for various coupling reactions and functional group transformations required in advanced drug discovery projects.
Quality Assurance and Safety
As a responsible manufacturer, we implement a comprehensive quality management system aligned with international pharmaceutical standards. Each production batch undergoes extensive testing using HPLC, NMR, and mass spectrometry to confirm identity and purity. We provide a detailed Certificate of Analysis (COA) with every shipment, ensuring full traceability and compliance. Our facility maintains strict environmental controls to prevent contamination, and all products are packaged in secure, moisture-resistant containers to preserve stability during global transit.
Storage and Handling
To maintain optimal chemical properties, this product should be stored in a cool, dry, and well-ventilated area. Containers must remain tightly closed when not in use to protect against moisture and strong light. Handling should be performed in accordance with standard laboratory safety protocols, including the use of appropriate personal protective equipment. Proper storage conditions ensure the material retains its specified purity and performance characteristics throughout its shelf life, supporting uninterrupted production schedules for our partners.



