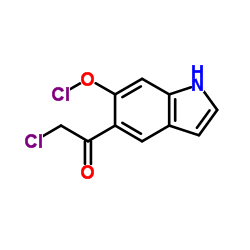
5-Chloroacetyl-6-chlorooxindole
- CAS No.118307-04-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate designed for Ziprasidone API synthesis. Reliable supply chain with comprehensive quality documentation for global manufacturers.
Request Bulk PricingProduct Technical Details
Product Overview
5-Chloroacetyl-6-chlorooxindole represents a critical structural motif in modern medicinal chemistry, specifically serving as a key building block for the synthesis of advanced pharmaceutical active ingredients. This oxindole derivative is engineered to meet the rigorous demands of API manufacturing, ensuring consistent performance in downstream chemical reactions. Our facility adheres to strict quality management systems to deliver materials that support efficient production cycles.
As a specialized chemical entity, this compound features a chloroacetyl group attached to the oxindole core, providing high reactivity for subsequent nucleophilic substitutions. The manufacturing process is optimized to minimize impurities, resulting in a product that facilitates smoother synthesis pathways for complex therapeutic agents. We prioritize batch-to-batch consistency to support large-scale industrial applications.
Technical Specifications
Quality control is paramount in pharmaceutical intermediate supply. Each batch undergoes comprehensive analytical testing to verify compliance with specified parameters. The following table outlines the standard physical and chemical properties associated with this material.
| Parameter | Specification |
|---|---|
| CAS Number | 118307-04-3 |
| Molecular Formula | C10H7Cl2NO2 |
| Molecular Weight | 244.07 g/mol |
| Appearance | Light Brown Solid |
| Purity | ≥98.0% |
| Melting Point | 202-206°C |
| Density | 1.471 g/cm3 |
| Boiling Point | 439.1°C at 760 mmHg |
Industrial Applications
This intermediate is primarily utilized in the pharmaceutical sector for the construction of antipsychotic medication frameworks. It serves as a essential precursor in the multi-step synthesis of Ziprasidone and its various salt forms. The high purity level ensures that downstream reactions proceed with optimal yield, reducing waste and processing time for API manufacturers.
- Compatible with standard organic synthesis protocols.
- Designed for scalability in industrial reactor settings.
- Supports the production of high-value therapeutic compounds.
- Validated for use in regulated pharmaceutical supply chains.
Storage and Handling
To maintain chemical stability and integrity, proper storage conditions are essential. This material should be kept in a cool, dry environment away from direct sources of heat and moisture. Containers must remain tightly sealed when not in use to prevent degradation or contamination from atmospheric humidity.
Standard packaging includes 25kg cardboard drums, though custom packaging solutions are available to meet specific logistical requirements. We recommend handling this compound with appropriate personal protective equipment in accordance with standard laboratory safety practices. Comprehensive Certificates of Analysis (COA) are provided with every shipment to verify quality standards.



