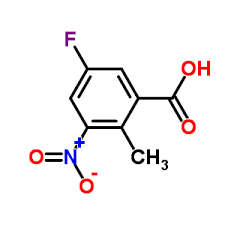
5-Fluoro-2-methyl-3-nitrobenzoic acid
- CAS No.850462-64-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity pharmaceutical intermediate for Rucaparib synthesis. Available in bulk quantities with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
5-Fluoro-2-methyl-3-nitrobenzoic acid represents a critical building block in modern medicinal chemistry, specifically designed for the synthesis of advanced pharmaceutical active ingredients. As a fluorinated aromatic compound, this intermediate plays a pivotal role in constructing complex molecular frameworks required for targeted cancer therapies. Our facility specializes in the large-scale production of this high-value chemical, ensuring consistent quality and supply chain reliability for global pharmaceutical manufacturers.
The incorporation of fluorine atoms into organic molecules often enhances metabolic stability and bioavailability, making this compound particularly valuable in drug discovery and development pipelines. We adhere to stringent manufacturing protocols to guarantee that every batch meets the rigorous demands of API synthesis, supporting the production of life-saving medications such as PARP inhibitors.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 5-Fluoro-2-methyl-3-nitrobenzoic acid |
| CAS Number | 850462-64-5 |
| Molecular Formula | C8H6FNO4 |
| Molecular Weight | 199.14 g/mol |
| Purity | ≥98.0% |
| Appearance | White to yellow solid |
Industrial Applications
This specialized intermediate is primarily utilized in the manufacturing process of Rucaparib, a potent poly ADP-ribose polymerase (PARP) inhibitor used in oncology treatments. Beyond this specific application, the chemical structure offers versatility for various organic synthesis routes requiring a fluorinated nitrobenzoic acid scaffold. Our clients leverage this material for research and development as well as commercial-scale production.
- Key intermediate for anticancer drug synthesis
- Essential building block for PARP inhibitor manufacturing
- Suitable for custom organic synthesis projects
- High stability for long-term storage and processing
Quality Assurance and Handling
Quality is the cornerstone of our operations. Each production lot undergoes comprehensive analytical testing using high-performance liquid chromatography (HPLC) and nuclear magnetic resonance (NMR) to verify identity and purity. We provide a detailed Certificate of Analysis (COA) with every shipment, ensuring full transparency and compliance with international pharmaceutical standards.
Proper storage is essential to maintain product integrity. We recommend storing this material in a cool, ventilated area away from direct sunlight and moisture. Our packaging solutions include 25 kg drums as standard, with flexible options available to meet specific logistical requirements. As a responsible global manufacturer, we ensure safe handling procedures are documented and provided to all partners to maintain workplace safety and environmental compliance.



