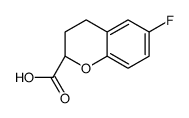
(2S)-6-Fluoro-2-chromanecarboxylic acid
- CAS No.129101-36-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity (2S)-6-Fluoro-2-chromanecarboxylic acid is a critical chiral intermediate used extensively in the synthesis of Nebivolol hydrochloride for cardiovascular applications.
Request Bulk PricingProduct Technical Details
Product Overview
(2S)-6-Fluoro-2-chromanecarboxylic acid is a specialized pharmaceutical intermediate characterized by its specific chiral configuration at the 2-position of the chromane ring. This compound serves as a fundamental building block in the modern synthesis of beta-blockers, specifically acting as a key precursor for Nebivolol hydrochloride. The presence of the fluorine substituent at the 6-position enhances the metabolic stability and binding affinity of the final pharmaceutical product, making this intermediate highly valuable in cardiovascular drug development.
Our manufacturing process ensures strict stereochemical control, guaranteeing the high enantiomeric excess required for downstream pharmaceutical applications. As a regulated chemical entity, this product is supplied exclusively for industrial synthesis and research purposes, adhering to international quality standards.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | (2S)-6-Fluoro-2-chromanecarboxylic acid |
| CAS Number | 129101-36-6 |
| Molecular Formula | C10H9FO3 |
| Molecular Weight | 196.18 g/mol |
| Appearance | White solid |
| Purity | ≥99.0% |
| Density | 1.364 g/cm3 |
| Boiling Point | 358.0 °C |
Industrial Applications
The primary application of this fluorinated chromane derivative is in the production of Nebivolol, a third-generation beta-blocker used to treat hypertension and heart failure. The (2S) enantiomer is crucial because the pharmacological activity of Nebivolol is highly stereoselective. Utilizing high-purity intermediates ensures the final drug product meets rigorous safety and efficacy profiles required by global health authorities.
Beyond Nebivolol synthesis, this compound is utilized in medicinal chemistry research for developing novel cardiovascular agents. Its unique structural features allow chemists to explore structure-activity relationships in benzopyran-based therapeutics. We support both large-scale commercial production and small-batch research requirements.
Quality Assurance and Storage
Quality control is paramount in our production facility. Each batch undergoes comprehensive analysis using HPLC, NMR, and mass spectrometry to verify identity, purity, and chiral integrity. A Certificate of Analysis (COA) is provided with every shipment to ensure traceability and compliance with your internal quality standards.
- Strict adherence to GMP-like manufacturing protocols.
- Regular auditing of raw material sources.
- Stability testing under various conditions.
For optimal stability, store this compound in a tightly closed container in a cool, dry, and well-ventilated area. Protect from moisture and direct sunlight. Keep away from incompatible substances such as strong oxidizing agents. Proper handling procedures should be followed according to standard laboratory safety guidelines.



