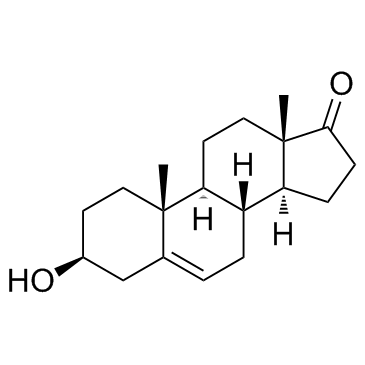
Dehydroepiandrosterone
- CAS No.53-43-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Dehydroepiandrosterone (CAS 53-43-0) designed for pharmaceutical synthesis. Ideal intermediate for Abiraterone production with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
Dehydroepiandrosterone, widely recognized in the chemical industry by its CAS registry number 53-43-0, stands as a critical steroid intermediate within the pharmaceutical manufacturing sector. This compound serves as a foundational building block for the synthesis of complex hormonal therapeutics and advanced medicinal agents. Our facility specializes in the production of this high-purity chemical, ensuring that every batch meets rigorous international standards for pharmaceutical intermediates. The molecular structure, characterized by its specific hydroxy and ketone functional groups, makes it an indispensable precursor in modern organic synthesis pathways.
As a leading global manufacturer, we understand the importance of consistency and reliability in the supply chain. Our Dehydroepiandrosterone is produced under strictly controlled conditions to minimize impurities and maximize yield. This commitment to quality ensures that downstream manufacturers can rely on our material for sensitive synthesis reactions without compromising the integrity of their final pharmaceutical products.
Chemical Properties and Stability
The physical and chemical characteristics of this intermediate are vital for its handling and processing in industrial settings. Typically presenting as a white crystalline powder, the compound exhibits excellent stability when stored under recommended conditions. The melting point range is tightly controlled between 149.0 and 151.0 degrees Celsius, serving as a key indicator of purity. With a molecular weight of approximately 288.42 g/mol, it possesses specific density and refractive index properties that are consistently verified during our quality assurance protocols.
Stability is further ensured through careful management of environmental factors such as moisture and light. The compound maintains its structural integrity when kept in well-closed containers away from strong heat sources. This robustness facilitates easier transportation and storage for our international clients, reducing the risk of degradation during logistics.
Quality Specifications
Quality control is the cornerstone of our manufacturing process. Each batch undergoes comprehensive analysis to verify compliance with factory standards and client requirements. The following table outlines the key specification parameters that define our premium grade material:
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | White crystalline powder | Conform |
| Assay | ≥99% | 99.5% |
| Melting Point | 149.0-151.0℃ | 150.0℃ |
| Loss on Drying | ≤0.5% | Conform |
| Heavy Metals | 10ppm Max | Conform |
| Conclusion | Conforms to Factory Standard | |
Industrial Applications
The primary application of this chemical lies in its role as a sophisticated intermediate for the synthesis of pharmaceutical agents. Specifically, it is utilized in the manufacturing process of Abiraterone and its acetate form, which are significant compounds in the field of oncology therapeutics. The high purity level of our intermediate ensures efficient reaction kinetics and reduces the burden on downstream purification steps.
Beyond specific drug synthesis, this compound is valuable for research and development laboratories focusing on steroid chemistry. Its reliable performance profile makes it a preferred choice for chemists developing new synthesis routes or optimizing existing manufacturing processes. We support our clients with technical data and documentation to facilitate regulatory filings and process validation.
Packaging and Storage
To maintain product integrity during transit, we offer flexible packaging solutions tailored to client needs. Standard packaging includes 25kg cardboard drums, which provide adequate protection against physical damage and environmental exposure. Custom packaging configurations are available upon request to accommodate specific logistical requirements or bulk orders.
Storage recommendations emphasize keeping the material in a cool, dry, and well-ventilated area. Containers should remain tightly sealed when not in use to prevent moisture absorption. By adhering to these storage guidelines, clients can ensure the long-term stability and usability of the chemical inventory. Our team is ready to assist with global shipping arrangements, ensuring timely delivery with full documentation including Certificates of Analysis.



