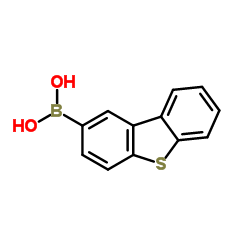
Dibenzothiophene-2-Boronic Acid
- CAS No.668983-97-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Dibenzothiophene-2-Boronic Acid designed for advanced OLED material synthesis, ensuring superior performance and reliability in electronic applications.
Request Bulk PricingProduct Technical Details
Product Overview
Dibenzothiophene-2-Boronic Acid stands as a pivotal chemical building block within the realm of advanced organic electronics. Specifically engineered for high-performance applications, this compound serves as a critical precursor in the manufacturing of organic light-emitting diode materials. Our production facility adheres to rigorous international quality standards, ensuring that every batch delivers consistent molecular integrity and exceptional purity levels required for sensitive electronic formulations.
The chemical structure features a robust dibenzothiophene core coupled with a reactive boronic acid functional group. This unique configuration facilitates efficient cross-coupling reactions, making it an indispensable reagent for chemists developing next-generation display technologies. By selecting our premium grade material, manufacturers can optimize their synthesis pathways while minimizing impurity-related defects in final products.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | Dibenzothiophene-2-Boronic Acid |
| CAS Number | 668983-97-9 |
| Molecular Formula | C12H9BO2S |
| Molecular Weight | 228.075 g/mol |
| Appearance | White Powder |
| Assay | ≥99.0% |
| Density | 1.38 g/cm3 |
| Boiling Point | 480.2°C |
| Flash Point | 244.2°C |
Industrial Applications
This specialized intermediate is predominantly utilized in the synthesis of complex OLED emissive layers and charge transport materials. The high purity assay ensures minimal quenching effects during device operation, thereby enhancing luminous efficiency and operational lifespan. Researchers and industrial chemists leverage this boronic acid derivative for Suzuki-Miyaura coupling reactions, enabling the precise construction of conjugated polymer systems essential for modern flat-panel displays and lighting solutions.
Beyond display technology, the compound finds utility in the development of organic photovoltaics and sensor technologies where stable sulfur-containing heterocycles are required. Its thermal stability and defined reactivity profile allow for scalable manufacturing processes without compromising on the electronic properties of the downstream materials. We support diverse formulation needs ranging from laboratory-scale research to full commercial production runs.
Quality Assurance And Safety
Quality control is paramount in our manufacturing process. Each production lot undergoes comprehensive analytical testing including HPLC, NMR, and mass spectrometry to verify structural identity and purity. We provide a detailed Certificate of Analysis with every shipment, ensuring full traceability and compliance with your internal quality protocols. Our facility implements strict environmental controls to prevent contamination during synthesis and packaging stages.
Regarding safety, this material should be handled in accordance with standard laboratory safety practices. It is recommended to use appropriate personal protective equipment including gloves and eye protection during handling. The compound exhibits stability under normal conditions but should be protected from moisture to maintain optimal reactivity. In case of spills, standard chemical containment procedures should be followed immediately to ensure workplace safety.
Packaging And Storage
To preserve chemical integrity during transit, Dibenzothiophene-2-Boronic Acid is packaged in secure 25 kg drums lined with moisture-resistant materials. Custom packaging solutions are available upon request to accommodate specific logistical requirements or smaller batch sizes. Storage recommendations include keeping the containers in a cool, well-ventilated area away from direct sunlight and incompatible substances. Proper sealing after each use is critical to prevent hydrolysis of the boronic acid group, ensuring the material remains ready for use in sensitive synthetic applications.



