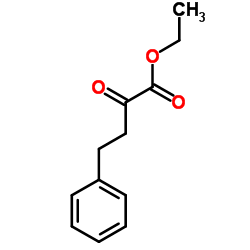
Ethyl 2-oxo-4-phenylbutyrate
- CAS No.64920-29-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Ethyl 2-oxo-4-phenylbutyrate (CAS 64920-29-2) supplied for pharmaceutical synthesis. Ideal intermediate for ACE inhibitors and antibacterial agents with verified COA.
Request Bulk PricingProduct Technical Details
Product Overview
Ethyl 2-oxo-4-phenylbutyrate is a specialized organic compound serving as a critical building block in the pharmaceutical industry. Recognized by its CAS registry number 64920-29-2, this substance functions primarily as a key intermediate in the multi-step synthesis of complex therapeutic agents. Our manufacturing facility produces this chemical under strict quality control protocols to ensure consistency and reliability for downstream pharmaceutical applications.
As a keto-ester derivative, this compound exhibits specific reactivity profiles that make it indispensable for constructing certain molecular frameworks required in cardiovascular and antibacterial medications. We prioritize the delivery of high-purity materials to support efficient reaction yields and minimize impurity profiles in final active pharmaceutical ingredients.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 64920-29-2 |
| Molecular Formula | C12H14O3 |
| Molecular Weight | 206.238 g/mol |
| Appearance | Light yellow oily liquid |
| Assay | ≥99.0% |
| Water Content | ≤0.1% |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 309.0±0.0 °C at 760 mmHg |
| Flash Point | 140.0±19.2 °C |
| Refractive Index | 1.506 |
Industrial Applications
This chemical intermediate is predominantly utilized in the synthesis of prominent pharmaceutical drugs. It serves as a vital precursor in the manufacturing process of Lisinopril, a widely prescribed antihypertensive agent belonging to the class of angiotensin-converting enzyme (ACE) inhibitors. Additionally, it finds application in the production of Cilastatin, an antibacterial drug enhancer used to protect certain antibiotics from enzymatic degradation.
Our supply chain supports large-scale pharmaceutical manufacturing needs. By providing material with stringent purity specifications, we help our partners maintain compliance with regulatory standards required for drug substance production. The consistency of our batch outputs ensures that synthesis routes remain robust and scalable.
Quality Assurance and Packaging
Every shipment of Ethyl 2-oxo-4-phenylbutyrate is accompanied by a comprehensive Certificate of Analysis (COA). Our quality assurance team verifies physical and chemical properties against internal standards before release. We understand the importance of material traceability in the pharmaceutical sector and maintain detailed records for every production lot.
Standard packaging consists of 25kg drums, designed to protect the integrity of the liquid during transit. Custom packaging solutions are available upon request to meet specific logistical requirements. We recommend storing the product in a cool, dry, and well-ventilated area with containers tightly closed to prevent moisture absorption or contamination.



