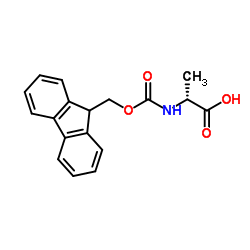
Fmoc-D-Ala-OH
- CAS No.79990-15-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Fmoc-D-Ala-OH (CAS 79990-15-1), a chiral building block for solid-phase peptide synthesis. Ideal for pharmaceutical R&D and GMP-compliant manufacturing.
Request Bulk PricingProduct Technical Details
Product Overview
Fmoc-D-Ala-OH, chemically designated as (2R)-2-(9H-fluoren-9-ylmethoxycarbonylamino)propanoic acid, is a high-value chiral amino acid derivative widely employed in the synthesis of complex peptides and peptidomimetics. As a D-configured alanine building block protected with the fluorenylmethyloxycarbonyl (Fmoc) group, it enables orthogonal deprotection strategies in modern solid-phase peptide synthesis (SPPS). This intermediate ensures excellent stereochemical integrity and coupling efficiency, making it indispensable in the development of therapeutic peptides, diagnostic agents, and advanced research compounds.
Specifications
| Molecular Formula | C₂₀H₁₇FO₆ |
|---|---|
| Molecular Weight | 372.34 g/mol |
| Appearance | White to off-white crystalline powder |
| Melting Point | 151–155 °C |
| Density | 1.3 ± 0.1 g/cm³ |
| Boiling Point | 544.1 ± 33.0 °C at 760 mmHg |
| Storage Conditions | Store in a sealed container at 2–8 °C in a cool, dry environment |
Quality Assurance
Our Fmoc-D-Ala-OH meets stringent quality benchmarks essential for pharmaceutical applications:
- Purity (HPLC): ≥98.5% (Typical: 99.54%)
- Chiral Purity: ≥99.0% (Typical: 99.92%)
- Water Content: ≤6.0% (Typical: 3.5%)
- Conforms to internal factory standards for use in regulated environments
Industrial Applications
Primarily utilized as a specialty intermediate in peptide chemistry, Fmoc-D-Ala-OH supports the construction of stereochemically defined sequences in:
- Solid-phase and solution-phase peptide synthesis
- Development of enzyme-resistant peptide therapeutics
- Preparation of chiral scaffolds for medicinal chemistry
- Academic and industrial research requiring enantiomerically pure D-amino acid units
As a foundational building block rather than a direct active ingredient, it plays a critical role in upstream synthesis workflows but is not intended for direct formulation into final consumer products.



