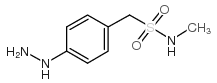
1-(4-Hydrazinylphenyl)-N-methylmethanesulfonamide
- CAS No.139272-29-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate for Sumatriptan synthesis. Available in bulk with full documentation.
Request Bulk PricingProduct Technical Details
Product Overview
1-(4-Hydrazinylphenyl)-N-methylmethanesulfonamide is a specialized organic compound serving as a critical building block in the pharmaceutical industry. Identified by CAS number 139272-29-0, this sulfonamide derivative plays an indispensable role in the multi-step synthesis of advanced therapeutic agents. Our facility produces this intermediate under strict quality control protocols to ensure consistency and reliability for downstream API manufacturing. The chemical structure combines a hydrazinyl phenyl group with a methylated sulfonamide moiety, offering unique reactivity profiles suitable for complex organic transformations.
As a dedicated manufacturer, we understand the stringent requirements of pharmaceutical supply chains. This product is engineered to meet high purity standards, minimizing impurities that could affect subsequent reaction yields or final drug safety. Whether used in research and development or large-scale commercial production, our material provides a stable foundation for synthesizing migraine treatment medications and related neurological therapeutics.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 1-(4-Hydrazinylphenyl)-N-methylmethanesulfonamide |
| CAS Number | 139272-29-0 |
| Molecular Formula | C8H13N3O2S |
| Molecular Weight | 215.27 g/mol |
| Purity | ≥98% |
| Appearance | Powder |
| Density | 1.356 g/cm3 |
| Boiling Point | 427.4°C at 760 mmHg |
| Flash Point | 212.3°C |
Industrial Applications
The primary application of this compound is as a key intermediate in the synthesis of Sumatriptan and its succinate salt. Sumatriptan is a widely recognized pharmaceutical active ingredient used globally for the acute treatment of migraine attacks. The precision of the intermediate directly influences the efficiency of the coupling reactions and the purity of the final API. Our production process optimizes the hydrazine functionality to ensure smooth progression through sulfonamide formation and subsequent cyclization steps.
Beyond specific API synthesis, this chemical serves as a valuable tool in medicinal chemistry laboratories for developing novel analogs. Researchers utilize this building block to explore structure-activity relationships within the triptan class of compounds. The stability of the sulfonamide group allows for diverse synthetic modifications, making it a versatile component in the discovery of new neurological therapeutics.
Quality Assurance and Storage
- Strict Quality Control: Every batch undergoes rigorous testing using HPLC and NMR to verify identity and purity levels.
- Documentation: Comprehensive COA, MSDS, and technical data sheets are provided with every shipment.
- Packaging: Available in 25kg cardboard drums or customized packaging based on client logistics requirements.
- Storage Conditions: Store in a cool, dry, well-closed container away from moisture and strong light or heat sources.
- Stability: When stored under recommended conditions, the product maintains its specified properties for extended periods.
We are committed to supplying global partners with reliable chemical solutions that adhere to international safety and quality standards. Our logistics network ensures timely delivery while maintaining product integrity during transit. For bulk inquiries or custom synthesis requests, our technical team is ready to assist with detailed specifications and competitive pricing structures tailored to your manufacturing needs.



