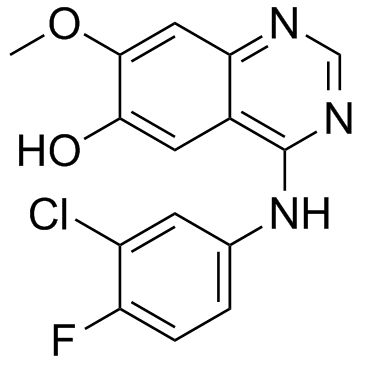
O-Desmorpholinopropyl Gefitinib
- CAS No.184475-71-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate essential for Gefitinib synthesis, offering superior stability and consistent quality for global API manufacturing.
Request Bulk PricingProduct Technical Details
Product Overview
O-Desmorpholinopropyl Gefitinib serves as a critical structural precursor in the pharmaceutical industry, specifically designed for the synthesis of advanced tyrosine kinase inhibitors. This quinazoline derivative represents a key building block in the production pipeline for targeted cancer therapeutics. Our manufacturing process ensures that each batch meets rigorous international standards, providing reliability for downstream API production.
As a specialized intermediate, this compound features a robust chemical structure that facilitates efficient coupling reactions. The precise molecular architecture allows for seamless integration into complex synthesis routes, minimizing side reactions and maximizing overall yield. We prioritize consistency in every production run to support large-scale pharmaceutical manufacturing requirements.
Technical Specifications
Quality control is paramount in pharmaceutical intermediate supply. Our facility utilizes advanced analytical methods to verify identity and purity. The following table outlines the standard physical and chemical parameters guaranteed for this product.
| Parameter | Specification |
|---|---|
| CAS Number | 184475-71-6 |
| Molecular Formula | C15H11ClFN3O2 |
| Molecular Weight | 319.72 g/mol |
| Appearance | Pale brown powder |
| Purity (HPLC) | ≥98.5% |
| Loss on Drying | ≤1.0% |
| Residue on Ignition | ≤0.1% |
| Melting Point | 260ºC |
Industrial Applications
This intermediate is primarily utilized in the research and development of epidermal growth factor receptor (EGFR) inhibitors. Pharmaceutical companies rely on high-quality precursors to ensure the safety and efficacy of the final active pharmaceutical ingredient. The compound's stability under standard storage conditions makes it suitable for global logistics and extended production schedules.
Beyond standard synthesis, this material supports process optimization studies. Chemists use this intermediate to refine reaction conditions, improve scalability, and reduce manufacturing costs. Our technical team provides comprehensive support to help clients integrate this material into their specific workflow efficiently.
Quality Assurance and Storage
We adhere to strict quality management systems to maintain product integrity. Each shipment is accompanied by a Certificate of Analysis (COA) detailing batch-specific test results. Proper storage is essential to maintain chemical stability over time. We recommend keeping the material in a cool, ventilated environment away from direct sunlight and moisture.
Packaging options are flexible to meet diverse client needs. Standard export drums ensure safety during transit, while customized packaging is available upon request. Our global supply chain capabilities ensure timely delivery to research centers and manufacturing facilities worldwide, supporting continuous production without interruption.



