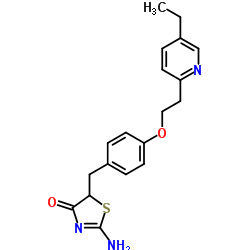
Pioglitazone 2-Imine
- CAS No.105355-26-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Pioglitazone 2-Imine intermediate designed for professional pharmaceutical API synthesis. Guaranteed assay ≥98% with comprehensive quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
Pioglitazone 2-Imine serves as a critical pharmaceutical intermediate in the synthesis of advanced antidiabetic active pharmaceutical ingredients. This specialized chemical building block is engineered to meet the rigorous demands of modern medicinal chemistry and large-scale API manufacturing. Our facility ensures that every batch undergoes strict quality control protocols to maintain consistency and reliability for downstream production processes.
As a key precursor, this compound plays an essential role in the construction of thiazolidinone-based therapeutic agents. The molecular structure is optimized for high reactivity and selectivity, facilitating efficient conversion into final drug substances. We prioritize purity and structural integrity, making this product an ideal choice for research laboratories and commercial pharmaceutical manufacturers alike.
Key Specifications
Quality assurance is paramount in pharmaceutical synthesis. Our Pioglitazone 2-Imine is produced under controlled conditions to minimize impurities and ensure optimal performance in subsequent reaction steps. The following table outlines the standard technical parameters verified by our analytical team.
| Parameter | Specification |
|---|---|
| CAS Number | 105355-26-8 |
| Molecular Formula | C19H21N3O2S |
| Molecular Weight | 355.454 g/mol |
| Appearance | Light yellow to light brown powder |
| Assay | ≥98.0% |
| Single Impurity | ≤1.0% |
| Melting Point | 187-188ºC |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 557.4±60.0 ºC at 760 mmHg |
Industrial Applications
This intermediate is primarily utilized in the manufacturing of Pioglitazone hydrochloride, a well-known therapeutic agent used in the management of type 2 diabetes. The high purity level ensures that the final API meets international pharmacopoeia standards. Beyond standard production, this chemical is valuable for process development, generic drug formulation, and analytical reference standard preparation.
Our clients leverage this material for scaling up synthesis routes from laboratory to pilot plant and full commercial production. The consistent quality reduces the risk of batch failures and optimizes overall yield in multi-step synthesis pathways. We support pharmaceutical companies in maintaining regulatory compliance through detailed documentation and traceability.
Storage and Handling
To preserve chemical stability and potency, proper storage conditions are essential. The product should be kept in a cool, ventilated area away from direct sunlight and moisture sources. Containers must remain tightly sealed when not in use to prevent contamination or degradation. Standard industrial safety precautions apply during handling, including the use of appropriate personal protective equipment.
We offer flexible packaging solutions, typically supplying in 25 kg drums, with customization available to meet specific logistical requirements. Global shipping capabilities ensure timely delivery to manufacturing sites worldwide. For detailed safety data sheets, certificates of analysis, or custom synthesis inquiries, our technical support team is ready to assist with your project needs.



