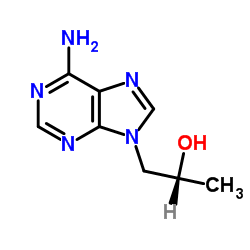
(R)-(+)-9-(2-Hydroxypropyl)Adenine
- CAS No.14047-28-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used primarily in the synthesis of antiviral agents. Manufactured under strict quality control standards for global B2B supply.
Request Bulk PricingProduct Technical Details
Product Overview
(R)-(+)-9-(2-Hydroxypropyl)Adenine is a specialized chiral pharmaceutical intermediate critical for the development and manufacturing of advanced nucleoside analogs. Our facility produces this compound with a strong emphasis on stereochemical purity, ensuring that the (R)-enantiomer is maintained at optimal levels for downstream synthesis processes. This chemical serves as a foundational building block in the pharmaceutical industry, particularly for researchers and manufacturers focused on antiviral therapeutic agents.
We understand the stringent requirements of modern drug synthesis. Therefore, our production lines are designed to minimize impurities and maximize yield consistency. Each batch undergoes rigorous analytical testing to confirm identity and potency before release. This commitment to excellence makes our product a preferred choice for global pharmaceutical companies seeking reliable supply chains for complex organic synthesis.
Technical Specifications
Quality parameters are strictly monitored to meet international standards. The following table outlines the key physical and chemical properties verified during our quality control process.
| Parameter | Specification |
|---|---|
| CAS Number | 14047-28-0 |
| Molecular Formula | C8H11N5O |
| Molecular Weight | 193.21 g/mol |
| Appearance | White to off-white crystalline powder |
| Assay (HPLC) | ≥97.0% |
| Loss on Drying | ≤2.0% |
| Total Impurities | ≤3.0% |
| Melting Point | 193°C |
Quality Control and Assurance
Our quality assurance protocol involves multi-stage testing using high-performance liquid chromatography (HPLC) and nuclear magnetic resonance (NMR) spectroscopy. We prioritize the detection of any stereoisomeric contaminants to ensure the optical purity required for sensitive biological applications. Documentation such as Certificates of Analysis (COA) and Method Validation Reports are provided with every shipment to facilitate regulatory compliance in your jurisdiction.
Continuous improvement is central to our manufacturing philosophy. We regularly audit our synthesis routes and purification methods to adopt the latest advancements in green chemistry and process efficiency. This approach not only ensures product stability but also supports sustainable manufacturing practices valued by our international partners.
Storage and Handling Guidelines
To maintain the integrity of (R)-(+)-9-(2-Hydroxypropyl)Adenine, proper storage conditions are essential. We recommend the following handling procedures:
- Store in a cool, ventilated warehouse away from direct sunlight.
- Keep containers tightly sealed to prevent moisture absorption.
- Maintain storage temperature below 25°C for long-term stability.
- Use appropriate personal protective equipment during handling and weighing.
- Avoid contact with strong oxidizing agents or incompatible materials.
Industrial Applications
This intermediate is predominantly utilized in the synthesis of nucleoside reverse transcriptase inhibitors. Its high purity profile makes it suitable for scaling up from laboratory research to commercial production volumes. By partnering with us, clients gain access to a stable supply of critical raw materials necessary for developing next-generation antiviral medications. We support bulk orders with flexible packaging options, including 25 kg drums or customized solutions based on specific logistical requirements.



