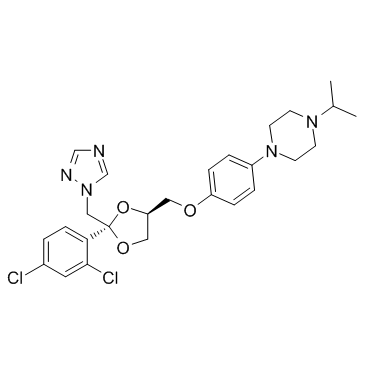
Terconazole
- CAS No.67915-31-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Terconazole intermediate for pharmaceutical manufacturing. Complies with strict quality standards for antifungal drug production.
Request Bulk PricingProduct Technical Details
Product Overview
Terconazole represents a critical component in the modern pharmaceutical landscape, specifically designed for use as a high-grade intermediate in the development of antifungal therapeutic agents. As a specialized chemical entity, this material is engineered to meet the rigorous demands of pharmaceutical manufacturing processes. Our facility ensures that every batch undergoes comprehensive analytical verification to guarantee consistency, purity, and reliability for downstream production applications.
The chemical structure of Terconazole is characterized by its specific molecular configuration, which contributes to its efficacy within formulated medicinal products. We prioritize the delivery of this substance with optimal physical properties, ensuring it integrates seamlessly into various pharmaceutical preparation workflows. Our commitment to excellence means that clients receive a product that aligns with international quality expectations for active pharmaceutical ingredients and intermediates.
Chemical Properties
Understanding the physicochemical characteristics of Terconazole is essential for proper handling and integration into manufacturing lines. The substance exhibits a defined molecular formula of C26H31Cl2N5O3, with a molecular weight of approximately 532.46 g/mol. These parameters are critical for stoichiometric calculations and quality control during the synthesis of final dosage forms.
The material typically presents as an off-white to white crystalline powder, indicating high levels of crystallinity and purity. Key physical constants include a melting point range centered around 126 degrees Celsius, which serves as a vital identifier for authenticity and purity verification. Additionally, the compound demonstrates specific density and refractive index values that are monitored during our internal quality assurance protocols to ensure batch-to-batch uniformity.
Specifications and Quality Data
Quality control is the cornerstone of our supply chain. We provide detailed certificates of analysis for every shipment, documenting key performance indicators against strict factory standards. The following table outlines the typical specification profile for our Terconazole intermediate:
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | Off-white to white crystalline powder | Conform |
| Related Substance (Single) | ≤0.25% | 0.14% |
| Related Substance (Total) | ≤0.5% | 0.18% |
| Assay | 98.0-102.0% | 99.86% |
| Melting Point | 125.0-129.0°C | 126.2-127.4°C |
| Loss on Drying | ≤1.0% | 0.08% |
| Residue on Ignition | ≤0.1% | 0.05% |
Industrial Applications
This pharmaceutical intermediate is primarily utilized in the manufacture of antifungal medications. It serves as a key active substance in various formulations intended for clinical use. Due to its specialized nature, handling should be restricted to qualified pharmaceutical manufacturing environments equipped with appropriate safety and containment measures. The material is suitable for incorporation into topical creams, suppositories, and other therapeutic delivery systems designed to address fungal infections.
Storage and Handling
To maintain the integrity of Terconazole, proper storage conditions are imperative. The material should be kept in its original sealed container within a cool, dark, and dry environment. Exposure to excessive heat, moisture, or direct sunlight should be avoided to prevent degradation. Adhering to these storage guidelines ensures that the chemical properties remain stable over time, preserving the value and usability of the product for future manufacturing cycles.
Quality Assurance and Compliance
Our production facilities operate under stringent quality management systems. We understand the importance of regulatory compliance in the pharmaceutical sector. Each batch is tested for impurities, assay content, and physical characteristics before release. We provide full documentation, including Certificates of Analysis (COA), to support our clients' regulatory filings and quality audits. Partnering with us ensures a reliable supply chain backed by technical expertise and a commitment to pharmaceutical-grade standards.



