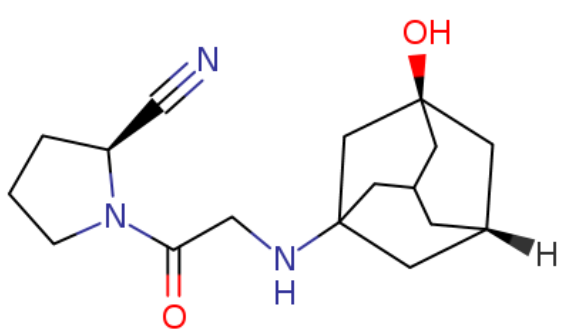
Vildagliptin
- CAS No.274901-16-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Vildagliptin API intermediate for type 2 diabetes formulations, offering stable supply and comprehensive quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
Vildagliptin represents a cornerstone in modern pharmaceutical development, specifically designed as a potent dipeptidyl peptidase-4 inhibitor. Our facility specializes in the production of high-purity Vildagliptin intermediates, catering to global pharmaceutical manufacturers seeking reliable supply chains for antidiabetic formulations. This compound is engineered to meet stringent quality standards, ensuring consistency across large-scale production batches. As a critical active pharmaceutical ingredient, Vildagliptin functions by enhancing the body's natural ability to lower blood sugar levels through enzymatic inhibition.
Our manufacturing process adheres to rigorous protocols to maintain chemical stability and potency, making it an ideal choice for downstream formulation partners. We prioritize transparency and technical support, providing detailed documentation to facilitate regulatory filings and quality control procedures for our international clients.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 274901-16-5 |
| Molecular Formula | C17H25N3O2 |
| Molecular Weight | 303.40 g/mol |
| Purity | ≥ 98% |
| Appearance | White Crystalline Powder |
| Density | 1.27 g/cm3 |
| Boiling Point | 531.3°C |
| Flash Point | 275.1°C |
Industrial Applications
The primary application of Vildagliptin lies in the synthesis of oral antidiabetic medications. It serves as a key component in therapies aimed at regulating glucose metabolism. Unlike some other agents, this substance offers a unique mechanism of action that inhibits glucagon secretion regardless of glycemic status, providing a balanced therapeutic profile. This characteristic makes it highly valuable for developing robust treatment regimens for patients with type 2 diabetes mellitus.
- Formulation of type 2 diabetes tablets and capsules.
- Development of combination therapies for metabolic disorders.
- Research and development in endocrinology pharmaceuticals.
- Generic drug manufacturing requiring high-purity API intermediates.
Quality Assurance and Supply
We understand the critical nature of supply chain reliability in the pharmaceutical sector. Every batch of Vildagliptin undergoes comprehensive analytical testing, including HPLC and mass spectrometry, to verify identity and purity. Certificates of Analysis (COA) are provided with every shipment to ensure full transparency and compliance with international regulatory expectations. Our quality management system is designed to trace every step of the production process.
Our logistics network supports global distribution, ensuring timely delivery while maintaining product integrity during transit. Whether for clinical trial materials or commercial-scale production, our team is equipped to handle diverse volume requirements with professionalism and discretion. We offer competitive bulk pricing structures for long-term partnerships.
Storage and Handling
To maintain optimal stability, Vildagliptin should be stored in a cool, dry place away from direct sunlight and moisture. Proper sealing of containers is essential to prevent degradation. Handling procedures should follow standard industrial hygiene practices, including the use of appropriate personal protective equipment. Adherence to these guidelines ensures the longevity and efficacy of the chemical substance throughout its shelf life.



