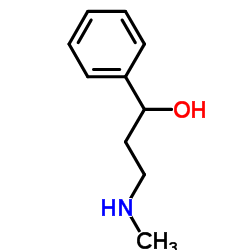
(1R)-3-(Methylamino)-1-phenylpropan-1-ol
- CAS No.115290-81-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used in the synthesis of ADHD therapeutics. Available in bulk with full documentation.
Request Bulk PricingProduct Technical Details
Product Overview
(1R)-3-(Methylamino)-1-phenylpropan-1-ol is a specialized chiral organic compound serving as a critical building block in modern pharmaceutical synthesis. Identified by CAS number 115290-81-8, this substance is primarily utilized in the manufacturing of neurological therapeutic agents. Our facility produces this intermediate under strict quality control protocols to ensure consistent stereochemical purity and chemical integrity. As a key precursor, it supports the development of medications designed for attention deficit disorders, adhering to global regulatory standards for pharmaceutical raw materials.
We understand the importance of reliability in the supply chain for active pharmaceutical ingredient (API) synthesis. Therefore, every batch undergoes rigorous analytical testing using advanced chromatography and spectroscopy methods. This ensures that the material meets the demanding specifications required by downstream manufacturers. Our commitment to excellence makes us a preferred partner for laboratories and production facilities seeking dependable sources of complex organic intermediates.
Technical Specifications
The following table outlines the standard physical and chemical properties associated with this product. Please note that specific parameters may vary slightly based on production batches, and a Certificate of Analysis (COA) is provided with every shipment.
| Parameter | Value |
|---|---|
| CAS Number | 115290-81-8 |
| Molecular Formula | C10H15NO |
| Molecular Weight | 165.23 g/mol |
| Appearance | White powder |
| Purity | ≥98.0% |
| Melting Point | 150-151 °C |
| Boiling Point | 286.1 °C at 760 mmHg |
| Density | 1.017 g/cm3 |
| Refractive Index | 1.53 |
Quality Assurance and Manufacturing
Our production process emphasizes safety, consistency, and environmental responsibility. We employ state-of-the-art synthesis routes to maximize yield while minimizing impurities. The chiral center at the (1R) position is carefully controlled to ensure optical purity, which is vital for the efficacy of the final pharmaceutical product. Our quality assurance team implements multiple checkpoints throughout the manufacturing process.
- Strict raw material sourcing from verified suppliers.
- In-process monitoring using HPLC and NMR techniques.
- Final batch release testing against international pharmacopoeia standards.
- Comprehensive documentation including COA, MSDS, and method validation reports.
Storage and Handling
To maintain stability and potency, this chemical intermediate should be stored in a cool, dry, and well-ventilated area. Containers must remain tightly closed to prevent exposure to moisture and strong light. Heat sources should be avoided to prevent degradation. We recommend storing the material in its original packaging until ready for use. Proper personal protective equipment (PPE) should be worn during handling to ensure safety in accordance with standard laboratory practices.
Industrial Applications
This compound is predominantly used as an intermediate in the synthesis of Atomoxetine hydrochloride. It serves as a fundamental structure for building more complex molecular architectures required in neurological therapeutics. Beyond specific API production, it is valuable in research and development settings focused on organic synthesis and medicinal chemistry. Our global logistics network ensures timely delivery to support continuous manufacturing operations worldwide.



