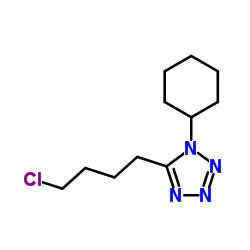
1-Cyclohexyl-5-(4-Chlorobutyl)-1H-Tetrazole
- CAS No.73963-42-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate for Cilostazol synthesis. Offers excellent stability and assay ≥99.0% for reliable API manufacturing.
Request Bulk PricingProduct Technical Details
Product Overview
1-Cyclohexyl-5-(4-Chlorobutyl)-1H-Tetrazole is a specialized organic compound serving as a critical building block in the pharmaceutical industry. Primarily utilized as a key intermediate in the synthesis of Cilostazol, this chemical plays an vital role in the production of cardiovascular therapeutic agents. Our manufacturing process ensures consistent quality and high purity levels, meeting the rigorous demands of modern API production facilities.
As a dedicated chemical supplier, we understand the importance of reliability in the supply chain. This tetrazole derivative is produced under strict quality control protocols to guarantee performance in downstream reactions. The structural integrity of the cyclohexyl and chlorobutyl moieties is maintained throughout production, ensuring optimal reactivity for pharmaceutical manufacturers.
Key Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 73963-42-5 |
| Molecular Formula | C11H19ClN4 |
| Molecular Weight | 242.748 |
| Appearance | White to off-white powder |
| Assay (HPLC) | ≥99.0% |
| Melting Point | 49-52°C |
| Moisture Content | ≤0.5% |
| Residue on Ignition | ≤0.2% |
Quality Assurance
Quality is the cornerstone of our chemical manufacturing operations. Each batch of 1-Cyclohexyl-5-(4-Chlorobutyl)-1H-Tetrazole undergoes comprehensive analytical testing to verify compliance with established standards. We employ advanced chromatographic and spectroscopic methods to confirm identity and purity.
- Identity confirmation via IR spectrum and HPLC retention time.
- Strict monitoring of related substances and individual impurities.
- Residual solvent analysis ensures safety and compliance with international guidelines.
- Certificate of Analysis (COA) provided with every shipment for traceability.
Our commitment to industrial purity means that clients can rely on consistent performance in their synthesis routes. Minimizing impurities is essential for maximizing yield in the final API production, and our refined manufacturing process is designed to achieve this goal efficiently.
Applications And Storage
This chemical is specifically designed for use as an intermediate in the pharmaceutical sector. It is not intended for direct consumer use or medical application without further processing. The primary application involves the multi-step synthesis of Cilostazol, where it serves as a foundational structure for the final active pharmaceutical ingredient.
Proper storage is essential to maintain the stability and quality of the product. We recommend storing the material in a cool, ventilated area away from direct sunlight and moisture. Containers should remain tightly sealed when not in use to prevent contamination or degradation. Our packaging options include 25 kg drums, with custom packaging available upon request to suit specific logistical requirements.
For global manufacturers seeking a reliable source of high-quality tetrazole intermediates, we offer competitive bulk pricing and robust logistical support. Contact our technical team for detailed information regarding synthesis routes, safety data, and volume discounts.



