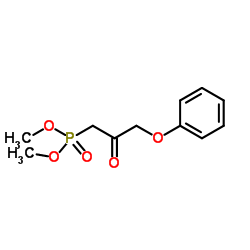
1-Dimethoxyphosphoryl-3-phenoxypropan-2-one
- CAS No.40665-68-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity pharmaceutical intermediate for prostaglandin analog synthesis. Reliable supply with comprehensive quality documentation for global manufacturing partners.
Request Bulk PricingProduct Technical Details
Product Overview
1-Dimethoxyphosphoryl-3-phenoxypropan-2-one is a specialized organic phosphonate compound designed for advanced pharmaceutical synthesis. This high-performance intermediate plays a critical role in the manufacturing of complex therapeutic agents, particularly within the prostaglandin analog class. Our facility produces this chemical under strict quality control protocols to ensure consistent molecular integrity and batch-to-batch reproducibility.
As a key building block, this phosphonate derivative offers superior reactivity and stability during multi-step synthesis routes. It is engineered to meet the rigorous demands of modern medicinal chemistry, providing chemists with a reliable foundation for constructing biologically active molecules. The compound is characterized by its distinct yellow oily liquid appearance and high assay purity, making it suitable for sensitive downstream reactions.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 40665-68-7 |
| Molecular Formula | C11H15O5P |
| Molecular Weight | 258.208 g/mol |
| Appearance | Yellow oily liquid |
| Assay | ≥98.0% |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 369.8±22.0 °C at 760 mmHg |
| Flash Point | 191.1±42.7 °C |
Industrial Applications
This chemical serves as a vital intermediate in the synthesis of specific prostaglandin analogs, including Etiproston and Tafluprost. These end products are widely utilized in ophthalmic and veterinary therapeutic applications. The phosphonate moiety within the structure facilitates specific carbon-carbon bond formations required for the biological activity of the final drug substance.
Our manufacturing process optimizes the yield and purity of this intermediate to minimize impurities that could affect downstream catalytic steps. By utilizing advanced purification techniques, we ensure that the material meets the stringent requirements of pharmaceutical grade synthesis. This reliability helps our partners reduce production risks and maintain efficient manufacturing timelines.
Quality Assurance and Storage
Quality is paramount in pharmaceutical intermediate production. Each batch undergoes comprehensive analytical testing, including HPLC and NMR verification, to confirm identity and purity levels. We provide a Certificate of Analysis (COA) with every shipment, detailing all critical quality attributes. Our packaging options include 200 kg drums, with flexibility to accommodate specific customer logistical requirements.
For optimal stability, store this product in a cool, ventilated area away from direct sunlight and moisture. Proper handling procedures should be followed to maintain the chemical properties during transportation and warehousing. Our global logistics network ensures timely delivery while maintaining the integrity of the product throughout the supply chain. Contact our technical sales team for detailed safety data sheets and bulk pricing information.



